The theory of Anthropogenic Global Warming, so far as we understand it, consists of the following two assertions.
(1) If we increase the concentration of CO2 in the atmosphere to 600 ppmv (parts per million by volume), we will cause the world to warm up by at least 2°C. (The concentration in pre-industrial times was 300 ppmv and is currently 400 ppmv.)
(2) If we continue burning fossil fuels at our current rate, emitting 10 petagrams of carbon into the atmosphere every year, we will raise the concentration of CO2 in the atmosphere to 600 ppmv within the next one hundred years.
We can falsify the second assertion using our observations of the carbon-14. We present a detailed analysis of atmospheric carbon-14 in a series of posts starting with Carbon-14: Origins and Reservoir. Here we present a summary, with approximate numerical values that are easy to remember.
Each year, cosmic rays create 8 kg of carbon-14 in the upper atmosphere. If carbon-14 were a stable atom, all carbon in the Earth's atmosphere would be carbon-14. But carbon-14 is not stable. One in eight thousand carbon-14 atoms decays each year. The rate at which the Earth's inventory of carbon-14 decays must be equal to the rate at which it is created. There must be 64,000 kg of carbon-14 on Earth.
The Earth's atmosphere contains 800 Pg of carbon (1 Pg = 1 Petagram = 1012 kg) bound up in gaseous CO2. One part per trillion of this carbon is carbon-14 (1 ppt = 1 part in 1012). There are 800 kg of carbon-14 in the atmosphere. That leaves 63,200 kg of the total inventory somewhere else. We'll call this "somewhere else" the carbon-14 reservoir.
Each year, 8 kg of carbon-14 is created in the atmosphere by cosmic rays, and each year the atmosphere loses 8 kg of carbon-14 to the reservoir. (Here we are ignoring the 0.1 kg of atmospheric carbon-14 that decays each year.) There is no chemical reaction that can separate carbon-14 from normal carbon. Every 1 kg of carbon-14 that leaves the atmosphere for the reservoir will be accompanied by 1 Pg of normal carbon.
Consider the atmosphere before we began to add 10 Pg of carbon to it each year. The mass of carbon in the atmosphere is constant. If 1 Pg of carbon leaves the atmosphere and enters the reservoir, 1 Pg of carbon must go in the opposite direction, leaving the reservoir and entering the atmosphere.
The only way for there to be a net loss of carbon-14 from the atmosphere to the reservoir is if the concentration of carbon-14 in the reservoir is lower than in the atmosphere. The only place on Earth that is capable of acting as the reservoir is the deep ocean, in which the concentration of carbon-14 is 80% of the concentration in the atmosphere. Each year 40 Pg of carbon leaves the atmosphere and enters the deep ocean, carrying with it 40 kg of carbon-14, while 40 Pg of carbon leaves the ocean and enters the atmosphere, carrying with it 32 kg of carbon-14. The result is a net flow of 8 kg/yr of carbon-14 into the ocean. Furthermore, the ocean contains 63,200 kg of carbon-14 in concentration 0.8 ppt, so the total mass of carbon in the oceans is roughly 80,000 Pg.
With the ocean and the atmosphere in equilibrium, 40 Pg of carbon is absorbed by the ocean each year, and 40 kg is released by the ocean. If we were to double the quantity of carbon in the atmosphere, we would double the amount absorbed by the ocean each year. Instead of 40 Pg being absorbed each year, 80 Pg would be absorbed. We could double the concentration of carbon in the atmosphere by emitting 40 Pg/yr. But we emit only 10 Pg/yr. Our emissions are sufficient to increase the mass of carbon in the atmosphere by 25%, after which everything we emit will be absorbed by the oceans. The oceans contain 80,000 Pg of carbon. If we add 10 Pg/yr, it will take roughly eight thousand years to double the carbon concentration in the oceans, after which the concentration in the atmosphere will double also.
Back in the 1960s, atmospheric nuclear bomb tests doubled the concentration of carbon-14 in the atmosphere. Such tests stopped in 1967. In our more precise calculation we predict that the concentration of carbon-14 must relax after 1967 with a time constant of 17 years, so that it would be 1.37 ppt in 1984 and 1.05 ppt in 2018. The concentration did relax afterwards, with a time constant of roughly 15 years, and in 2016, the carbon-14 concentration in the atmosphere is indistinguishable from its value before the bomb tests. During that time, almost every CO2 molecule that existed in the atmosphere in 1967 passed into the ocean and was replaced by another from the ocean. Anyone claiming that our carbon emissions will remain in the atmosphere for thousands of years, such as the author of this article, is wrong. If we stopped burning fossil fuels tomorrow, the CO2 concentration of the atmosphere would return to its pre-industrial value within fifty years.
When carbon is absorbed or emitted by the ocean, it does so as a molecule of CO2. Statistical mechanics dictates that the rate of absorption is weakly dependent upon temperature, but the rate of emission is strongly dependent upon temperature. When we calculate the effect of temperature upon the equilibrium between the ocean and the atmosphere, we conclude that a 1°C warming of the oceans will cause a 10 ppmv increase in the concentration of CO2 in the atmosphere. When we look back at the record of CO2 concentration and temperature over the past 400,000 years, we see the correlation we expect, with the magnitude of the changes in good agreement with our prediction. For a 12°C increase in temperature, for example, the concentration of CO2 increases by 110 ppmv.
If we consider the atmosphere of the Earth in pre-industrial times, its atmospheric CO2 concentration was roughly 300 ppmv. A more exact value for the creation of carbon-14 is 7.5 kg/yr and we conclude that 37 Pg/yr or carbon was being absorbed and emitted by the ocean. When we add 10 Pg/yr human emissions from burning fossil fuels, we expect the concentration of CO2 in the atmosphere to rise by 27% to 380 ppmv, which is close to the 400 ppmv we observe.
Our analysis of the carbon cycle makes three independent and unambiguous predictions all of which turn out to be correct to within ±10%. Our analysis is reliable, and it tells us that it will take roughly eight thousand years to double the CO2 concentration of the atmosphere if we continue burning fossil fuels at our current rate. Assertion (2) above is wrong by two orders of magnitude. The theory of Anthropogenic Global Warming, as stated above, is untrue.
POST SCRIPT: Assertion (1) is harder to falsify, and we do not claim to have done so in a manner convincing to all readers. Nevertheless, we did conclude that assertion (1) had to be wrong in our series of posts on the greenhouse effect, which we summarize in Anthropogenic Global Warming. We calculated that doubling the CO2 concentration of the atmosphere will cause the Earth to warm up by 1.5°C, provided we ignore changes in water vapor and cloud cover. As the world warms up, however, water evaporates more quickly from the oceans, and we get more clouds. Clouds reflect sunlight. The warming effect of doubling CO2 concentration is reduced by an increase in cloud cover. Clouds stabilize the Earth's temperature because they become more frequent as the Earth warms up, and less frequent as it cools down. Our simulation of the atmosphere with clouds suggests that the actual warming caused by a doubling of CO2 will be 0.9°C. So far as we can tell, the climate models used by the majority of climate scientists do not account for the increase in cloud cover that occurs as the world warms up. But they do account for the increase in water vapor in the atmosphere. Clouds cool the world, but water vapor is another greenhouse gas, and warms the world. By including water vapor but excluding the increasing cloud cover, these climate models conclude that the effect of doubling CO2 concentration will be 2°C or larger.
POST POST SCRIPT: Some readers suggest that the atmosphere-ocean system cannot be modeled with linear diffusion because the dissolved CO2 does not increase in proportion to atmospheric CO2 concentration. We address and reject their claim in an update to Probability of Exchange. They claim that Henry's Law does not apply to CO2 and seawater, despite many measurements to the contrary, such as Tsui et al., and general acceptance of Henry's Law in academic texts.
Showing posts with label Greenhouse Effect. Show all posts
Showing posts with label Greenhouse Effect. Show all posts
Sunday, October 9, 2016
Wednesday, April 4, 2012
Conclusion
The anthropogenic global warming (AGW) hypothesis presented by the majority of today's climatologists has two parts. First it claims that the world is getting exceptionally warm, and second it claims that human carbon dioxide (CO2) emissions are the cause of this warming. Seven years ago, we began our personal investigation of this hypothesis, and we did so by considering whether or not the world was indeed getting exceptionally warm.
The first thing we did was estimate the uncertainty inherent in the measurements of global surface temperature. We concluded that natural variations in local climate introduce an error of roughly 0.14°C in the measurement of the change in temperature between any two points in time. The fact that the error is constant with the time over which we measure the change is a consequence of the particular characteristics of local climate fluctuations.
We downloaded the weather station data from NCDC and calculated the global surface anomaly using a method we called integrated derivatives, but which others have called first differences. The graph we obtained was almost identical to the one obtained by CRU using their complex reference grid method. It remains a mystery to us why institutions like CRU, NASA, and NCDC use such a complex method when a far simpler one will do. All graphs show roughly a 0.6°C rise in global surface temperature from 1950 to 2000. This rise is significant compared to our expected resolution of 0.14°C.
We made this plot superimposing the number of weather stations and the global surface anomaly versus time. The number of weather stations drops dramatically from 1960 and 1990. Only one in four remain active at the end of this thirty-year period. During the same period, the global surface anomaly shows a 0.6°C rise. By selecting subsets of the weather stations, we found that the apparent warming from 1950 to 1990 varied from 0.3°C to 1.0°C depending upon whether we used stations that disappeared in that period, persisted through that period, or existed shorter or longer intervals in the same century. Thus is seemed to us that some significant amount of work would have to be done to eliminate the change in the number of weather stations as a source of error in the data. But we saw no mention whatsoever of this source of error in published papers in which the global surface anomaly is presented, such as Jones et al..
We plotted a global map of the available weather stations, color-coded to show the date they first started reporting. The map shows that almost all stations in the tropics began operating after 1930, while most of those in the temperate regions were operating by 1880. This seems to us to be another source of systematic error in our measurement of the global surface anomaly.
Weather stations might also be affected by the appearance of buildings, tarmac, and road traffic. We found examples of weather stations in which such urban heating caused an apparent warming of several degrees centigrade over a few decades. It seemed to us that this effect would have to be examined in depth by any paper presenting a global surface trend. But papers such as Jones et al. do not address the urban heating issue directly. Instead, they claim that the effect is negligible and refer to other papers as proof. But when we looked up those other papers, we did not find any such proof.
In order to argue that modern temperatures were exceptionally warm, climatologists produced the hockey stick graph, in which a collection of potential long-term measurements of global surface temperature were combined together under the assumption that they could be trusted only to the extent that they showed a temperatures increase from 1950 to 2000. Indeed, if a measurement showed a temperature decline in that period, the hockey stick method would flip the trend over and add it to the combination so that it now contributed to a rise in the same period.
The hockey-stick graph shows no sign of the Medieval Warm Period, in which Greenland was inhabited by farmers, nor the Little Ice Age, when the Thames was known to freeze over, and nor should we expect it to. Given a random set of measurements, the hockey-stick combination method will almost always produce a graph that shows a sharp rise from 1950 to 2000 and a gentle descent during the thousand years before-hand. When applied to the existing measurements of temperature by tree rings, ice cores, and other such indirect methods, it is no surprise that the method produced that same shape.
We presented our doubts about the surface temperature measurements and the hockey stick graph to believers in the AGW hypothesis. We were received with disdain and given no satisfactory answers. Furthermore, the Climategate affair revealed several significant breaches of scientific method by the climate science community. For example, in this graph produced by climatologists for the World Health Organization, the authors removed the tree ring temperature data from 1960 onwards because it showed a decline in temperature, and substituted temperature station measurements in their place. They plotted the combination as a single line. When I asked a prominent climatologists what exactly had been done, he said, "The smooth was calculated using instrumental data past 1960." He declared that a better way to handle the divergence of the tree-ring data from the station measurements would be to cut short the graph of tree-ring data at 1960, so as to hide the decline in temperatures measured by the tree rings.
What we see here is the assumption by climatologists that the world has been warming up and that the global temperature measured by weather stations is correct. This assumption leads them to delete conflicting data on the grounds that it must be bad data. Thus it becomes impossible for them to discover that their assumption is incorrect. By this time, we were skeptical of the global surface anomaly we obtained from the station data. We were no longer certain that the data itself had not been modified by NCDC. We had little reason to trust any other measurement produced by climatologists, we were unimpressed with the hockey-stick method of combining measurements, and we were quite certain that recent temperatures were not exceptional for the past ten thousand years.
We turned our attention to the second part of the AGW hypothesis: the one that says doubling the atmosphere's CO2 concentration will increase the surface temperature by roughly 3°C. It took us a long time to come to a conclusion on this one. The climate models upon which such predictions are based are private property of various climatologists. In any event, we do not trust models produced by a community that is willing to delete data that conflict with its assumptions. If they are willing to delete data, we must assume that they are willing to adjust their models until the models give predictions consistent with their AGW hypothesis.
We began with some laboratory experiments on radiation. We stated the principle of the greenhouse effect. After a great deal of searching around, we eventually obtained the absorption spectrum of various layers of the Earth's atmosphere. This allowed us to confirm that, if the skies remained clear, a doubling of CO2 concentration would cause the world to warm up by about 1.5°C.
But of course the skies don't remain clear. The formation of clouds is a strong function of surface temperature. If the world warms up, there will be more clouds. They will reflect more of the Sun's light, while at the same time, slowing down the radiation of heat into space by the Earth. To determine how these two effects would interact, we built our own climate model, which we called Circulating Cells.
When it comes to determining the effect of increased cloud cover, the most critical parameter to decide upon is the reflection of sunlight by clouds per millimeter of water depth in the cloud. It seemed to us that there should be a large body of literature written recently upon this subject because it is so important to climate modeling. The best paper we found upon the subject was written in 1948, Reflection, Absorption, and Transmission of Insolation by Stratus Cloud. We found a couple of more recent papers about reflection, such as this one, but they do not attempt to provide an empirical formula for the reflection of clouds with increasing cloud depth. We concluded that climatologists are not examining this issue in detail.
In a long sequence of small steps, we built up our climate model until it implemented surface convection, surface heat capacity, evaporation, cloud formation, precipitation, and radiation by clouds. We tested every aspect of the simulation in detail, and based its operating parameters upon our own estimates and upon whatever measurements we could find in climate science journals. We did not choose our model parameters to suit any hypothesis of our own, nor could we have done, because we did not have a model capable of testing the AGW hypothesis until the final stage, and we did not change the parameters in that final stage.
The latest version of our climate model shows that cloud cover increases rapidly as the surface warms above the freezing point of water. The evaporation rate of water from the surface increases approximately as the square of the temperature above freezing, and the only way for water to return to the surface is to form a cloud first. If we ignore the increased reflection of sunlight due to increasing cloud cover, and consider only the slowing-down of radiation into space by the same increase in cloud cover, our model shows roughly 3°C of warming due to a doubling in CO2 concentration. But when we take account of the increased reflection of sunlight by the increasing cloud cover, the warming drops to 0.9°C.
It seems to us that the climate models used by climatologists ignore the reflection of sunlight due to clouds. They may allow for some fixed fraction of sunlight to be reflected by clouds, but they do not allow this fraction to increase with increasing surface temperature. Thus they conclude that the warming due to CO2 doubling will be 3°C. If they took account of the increased reflection, the effect would be far smaller and less dramatic: roughly 1°C.
Doubling the CO2 concentration of the atmosphere will indeed encourage the world to warm up, but not by enough that we should worry. Right now CO2 concentration has increased from roughly 300 ppm to 400 ppm in the past century. If it gets to 600 ppm then we can say that the rise in CO2 concentration will tend to warm the Earth by 1°C. But we are unlikely to be able to check our calculations, because the natural variation in the Earth's climate is itself of order ±1°C from one century to the next.
And so we find ourselves at the end of our journey. Modern warming is not exceptional, and doubling the CO2 concentration will cause the world to warm up by roughly 1°C, not 3°C. The only part of the AGW theory we have not investigated is its assertion that human CO2 emissions are responsible for the increase in atmospheric CO2 concentration over the past century.
My thanks to those of you who took part in the effort, both by private e-mails and in the comments. I would not have continued the effort without your participation. I hope it is clear that my use of "we" instead of "I" is in recognition of the fact that this has been a group effort. I will continue to answer comments on this site, and I will consider any suggestions of further work. To the first approximation, however: we're done.
The first thing we did was estimate the uncertainty inherent in the measurements of global surface temperature. We concluded that natural variations in local climate introduce an error of roughly 0.14°C in the measurement of the change in temperature between any two points in time. The fact that the error is constant with the time over which we measure the change is a consequence of the particular characteristics of local climate fluctuations.
We downloaded the weather station data from NCDC and calculated the global surface anomaly using a method we called integrated derivatives, but which others have called first differences. The graph we obtained was almost identical to the one obtained by CRU using their complex reference grid method. It remains a mystery to us why institutions like CRU, NASA, and NCDC use such a complex method when a far simpler one will do. All graphs show roughly a 0.6°C rise in global surface temperature from 1950 to 2000. This rise is significant compared to our expected resolution of 0.14°C.
We made this plot superimposing the number of weather stations and the global surface anomaly versus time. The number of weather stations drops dramatically from 1960 and 1990. Only one in four remain active at the end of this thirty-year period. During the same period, the global surface anomaly shows a 0.6°C rise. By selecting subsets of the weather stations, we found that the apparent warming from 1950 to 1990 varied from 0.3°C to 1.0°C depending upon whether we used stations that disappeared in that period, persisted through that period, or existed shorter or longer intervals in the same century. Thus is seemed to us that some significant amount of work would have to be done to eliminate the change in the number of weather stations as a source of error in the data. But we saw no mention whatsoever of this source of error in published papers in which the global surface anomaly is presented, such as Jones et al..
We plotted a global map of the available weather stations, color-coded to show the date they first started reporting. The map shows that almost all stations in the tropics began operating after 1930, while most of those in the temperate regions were operating by 1880. This seems to us to be another source of systematic error in our measurement of the global surface anomaly.
Weather stations might also be affected by the appearance of buildings, tarmac, and road traffic. We found examples of weather stations in which such urban heating caused an apparent warming of several degrees centigrade over a few decades. It seemed to us that this effect would have to be examined in depth by any paper presenting a global surface trend. But papers such as Jones et al. do not address the urban heating issue directly. Instead, they claim that the effect is negligible and refer to other papers as proof. But when we looked up those other papers, we did not find any such proof.
In order to argue that modern temperatures were exceptionally warm, climatologists produced the hockey stick graph, in which a collection of potential long-term measurements of global surface temperature were combined together under the assumption that they could be trusted only to the extent that they showed a temperatures increase from 1950 to 2000. Indeed, if a measurement showed a temperature decline in that period, the hockey stick method would flip the trend over and add it to the combination so that it now contributed to a rise in the same period.
The hockey-stick graph shows no sign of the Medieval Warm Period, in which Greenland was inhabited by farmers, nor the Little Ice Age, when the Thames was known to freeze over, and nor should we expect it to. Given a random set of measurements, the hockey-stick combination method will almost always produce a graph that shows a sharp rise from 1950 to 2000 and a gentle descent during the thousand years before-hand. When applied to the existing measurements of temperature by tree rings, ice cores, and other such indirect methods, it is no surprise that the method produced that same shape.
We presented our doubts about the surface temperature measurements and the hockey stick graph to believers in the AGW hypothesis. We were received with disdain and given no satisfactory answers. Furthermore, the Climategate affair revealed several significant breaches of scientific method by the climate science community. For example, in this graph produced by climatologists for the World Health Organization, the authors removed the tree ring temperature data from 1960 onwards because it showed a decline in temperature, and substituted temperature station measurements in their place. They plotted the combination as a single line. When I asked a prominent climatologists what exactly had been done, he said, "The smooth was calculated using instrumental data past 1960." He declared that a better way to handle the divergence of the tree-ring data from the station measurements would be to cut short the graph of tree-ring data at 1960, so as to hide the decline in temperatures measured by the tree rings.
What we see here is the assumption by climatologists that the world has been warming up and that the global temperature measured by weather stations is correct. This assumption leads them to delete conflicting data on the grounds that it must be bad data. Thus it becomes impossible for them to discover that their assumption is incorrect. By this time, we were skeptical of the global surface anomaly we obtained from the station data. We were no longer certain that the data itself had not been modified by NCDC. We had little reason to trust any other measurement produced by climatologists, we were unimpressed with the hockey-stick method of combining measurements, and we were quite certain that recent temperatures were not exceptional for the past ten thousand years.
We turned our attention to the second part of the AGW hypothesis: the one that says doubling the atmosphere's CO2 concentration will increase the surface temperature by roughly 3°C. It took us a long time to come to a conclusion on this one. The climate models upon which such predictions are based are private property of various climatologists. In any event, we do not trust models produced by a community that is willing to delete data that conflict with its assumptions. If they are willing to delete data, we must assume that they are willing to adjust their models until the models give predictions consistent with their AGW hypothesis.
We began with some laboratory experiments on radiation. We stated the principle of the greenhouse effect. After a great deal of searching around, we eventually obtained the absorption spectrum of various layers of the Earth's atmosphere. This allowed us to confirm that, if the skies remained clear, a doubling of CO2 concentration would cause the world to warm up by about 1.5°C.
But of course the skies don't remain clear. The formation of clouds is a strong function of surface temperature. If the world warms up, there will be more clouds. They will reflect more of the Sun's light, while at the same time, slowing down the radiation of heat into space by the Earth. To determine how these two effects would interact, we built our own climate model, which we called Circulating Cells.
When it comes to determining the effect of increased cloud cover, the most critical parameter to decide upon is the reflection of sunlight by clouds per millimeter of water depth in the cloud. It seemed to us that there should be a large body of literature written recently upon this subject because it is so important to climate modeling. The best paper we found upon the subject was written in 1948, Reflection, Absorption, and Transmission of Insolation by Stratus Cloud. We found a couple of more recent papers about reflection, such as this one, but they do not attempt to provide an empirical formula for the reflection of clouds with increasing cloud depth. We concluded that climatologists are not examining this issue in detail.
In a long sequence of small steps, we built up our climate model until it implemented surface convection, surface heat capacity, evaporation, cloud formation, precipitation, and radiation by clouds. We tested every aspect of the simulation in detail, and based its operating parameters upon our own estimates and upon whatever measurements we could find in climate science journals. We did not choose our model parameters to suit any hypothesis of our own, nor could we have done, because we did not have a model capable of testing the AGW hypothesis until the final stage, and we did not change the parameters in that final stage.
The latest version of our climate model shows that cloud cover increases rapidly as the surface warms above the freezing point of water. The evaporation rate of water from the surface increases approximately as the square of the temperature above freezing, and the only way for water to return to the surface is to form a cloud first. If we ignore the increased reflection of sunlight due to increasing cloud cover, and consider only the slowing-down of radiation into space by the same increase in cloud cover, our model shows roughly 3°C of warming due to a doubling in CO2 concentration. But when we take account of the increased reflection of sunlight by the increasing cloud cover, the warming drops to 0.9°C.
It seems to us that the climate models used by climatologists ignore the reflection of sunlight due to clouds. They may allow for some fixed fraction of sunlight to be reflected by clouds, but they do not allow this fraction to increase with increasing surface temperature. Thus they conclude that the warming due to CO2 doubling will be 3°C. If they took account of the increased reflection, the effect would be far smaller and less dramatic: roughly 1°C.
Doubling the CO2 concentration of the atmosphere will indeed encourage the world to warm up, but not by enough that we should worry. Right now CO2 concentration has increased from roughly 300 ppm to 400 ppm in the past century. If it gets to 600 ppm then we can say that the rise in CO2 concentration will tend to warm the Earth by 1°C. But we are unlikely to be able to check our calculations, because the natural variation in the Earth's climate is itself of order ±1°C from one century to the next.
And so we find ourselves at the end of our journey. Modern warming is not exceptional, and doubling the CO2 concentration will cause the world to warm up by roughly 1°C, not 3°C. The only part of the AGW theory we have not investigated is its assertion that human CO2 emissions are responsible for the increase in atmospheric CO2 concentration over the past century.
My thanks to those of you who took part in the effort, both by private e-mails and in the comments. I would not have continued the effort without your participation. I hope it is clear that my use of "we" instead of "I" is in recognition of the fact that this has been a group effort. I will continue to answer comments on this site, and I will consider any suggestions of further work. To the first approximation, however: we're done.
Thursday, March 15, 2012
Anthropogenic Global Warming
The Anthropogenic Global Warming (AGW) hypothesis states that doubling the CO2 concentration of the Earth's atmosphere will raise the average surface temperature of the Earth by a minimum of 1.5°C, and more likely 3°C.
In our investigation of the absorption and emission of long-wave radiation by the Earth's atmosphere, we calculated that a sudden doubling of CO2 concentration would decrease the power the Earth radiates into space by 6.6 W/m2. We then estimated how much the Earth and its atmosphere would have to warm up in order to restore the heat radiated into space to its original value. We assumed there would be no significant change in cloud cover as a result of the warming, and we applied Stefan's Law to calculate how the heat radiated by the Earth and its atmosphere would increase. We found that the required increase in surface temperature would be around 1.6°C. If there were no change in cloud cover, then the heat arriving from the Sun would remain the same, and we could expect the Earth to warm up by 1.6°C so as to once again arrive at thermal equilibrium.
The AGW hypothesis states that in the event of the Earth warming, changes in cloud cover will be such as to amplify the warming we calculate using Stefan's Law. Here is an extract from today's entry on Global Warming at Wikipedia.
The main positive feedback in the climate system is the water vapor feedback. The main negative feedback is radiative cooling through the Stefan–Boltzmann law, which increases as the fourth power of temperature.
As the Earth warms up, water evaporates more quickly from the oceans. Almost all water that evaporates must turn into clouds before it returns to Earth. Water condensing directly onto grass in the morning is an exception to this rule, but the vast majority of water vapor will return only as rain or snow, and so must first take the form of a cloud.
Clouds absorb long-wave radiation emitted by the Earth's surface, so the Earth cannot radiate its heat directly into space. Instead, the clouds radiate into space and warm the Earth with back radiation. Thus increasing cloud cover means less heat radiated into space for the same surface temperature. This is the positive feedback referred to by the AGW hypothesis. The AGW climate models predict that this positive feedback will amplify the minimum 1.5°C warming caused by CO2 to roughly 3°C. Some say 2°C and other say 4°C, but all agree that the actual warming will be greater than 1.5°C.
We see this positive feedback in our Circulating Cells simulation, version CC11, which simulates the formation of clouds as well as their absorption and emission of long-wave radiation. The graph below shows a close-up of the behavior of the simulation in the neighborhood of its equilibrium point for 350 W/m2 solar power.
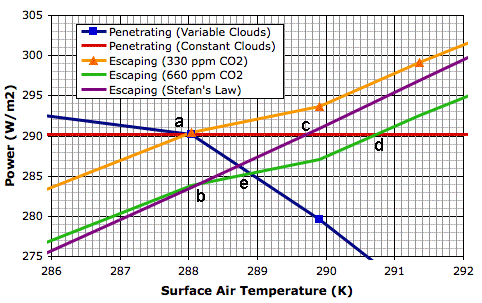
The blue line shows how the power that penetrates to the surface of our simulated planet varies with increasing surface temperature. The orange line shows how the total power escaping from our simulated planet increases with surface temperature. These two lines cross at a, where temperature is 288 K and total escaping power is 290 W/m2. Our simulated atmosphere absorbs 50% of long-wave radiation, which is an adequate approximation of our atmosphere with its current concentration of CO2 (roughly 330 ppm).
The green line is the same as the orange line, but displaced down by 6.6 W/m2, which is the amount by which we calculated the total power escaping from the Earth will decrease if we double CO2 concentration (to roughly 660 ppm). Thus the green line tells us the total escaping power at the same temperature if we were to double the CO2 concentration. Point b on the green line is 288 K, and the total escaping power is 283.4 W/m2.

The purple line shows how the total escaping power will increase from b if we assume the cloud cover is constant and use only Stefan's Law to determine the heat radiated into space by the surface and atmosphere. The red line shows the solar power penetrating to the surface if we assume the cloud cover is constant. With constant cloud cover, the penetrating solar power does not change.
The purple and red lines meet at c, which is 289.6 K, or 1.6°C above the previous equilibrium point. Thus our simulation shows us that the warming due to CO2 doubling, if we ignore changes in cloud cover, will be 1.6°C, which is consistent with our previous calculation.
The green line, however, is the simulation's calculation of the total escaping power for increasing surface temperature. We see that the heat radiated into space does not increase as quickly as Stefan's Law would lead us to expect. And the reason for that is precisely the reason quoted by the AGW hypothesis: increasing cloud cover is slowing down the radiation of heat into space. The green line and the red line intersect at d, which is 290.7 K, or 2.7°C above our original equilibrium temperature. This is the new equilibrium temperature of the planet surface if we double CO2 concentration and we assume that there will be no change in the solar power penetrating to the surface while the cloud cover increases.

But the solar power penetrating to the surface must decrease as cloud cover increases. Clouds reflect sunlight. Thick clouds reflect 90% of solar power back into space. Even thin, high clouds reflect 10%. Increasing cloud cover will decrease the solar power penetrating to the surface. That is why our blue line slopes downwards. This is negative feedback, which acts against the positive feedback described by the AGW theory. The blue line shows how the solar power penetrating to the surface decreases as our cloud cover increases.
The blue line and the green line intersect at e, which is the equilibrium point we arrive at after doubling the CO2 concentration and considering both the positive feedback of back-radiation and the negative feedback of solar reflection. The temperature at e is 288.9 K, which is 0.9°C above our original equilibrium temperature.
Thus our simulation shows how the negative feedback generated by clouds dominates their positive feedback, and suggests that the actual warming of the Earth's surface due to a doubling of CO2 will be closer to 0.9°C than the 3°C predicted by the AGW hypothesis.
In our investigation of the absorption and emission of long-wave radiation by the Earth's atmosphere, we calculated that a sudden doubling of CO2 concentration would decrease the power the Earth radiates into space by 6.6 W/m2. We then estimated how much the Earth and its atmosphere would have to warm up in order to restore the heat radiated into space to its original value. We assumed there would be no significant change in cloud cover as a result of the warming, and we applied Stefan's Law to calculate how the heat radiated by the Earth and its atmosphere would increase. We found that the required increase in surface temperature would be around 1.6°C. If there were no change in cloud cover, then the heat arriving from the Sun would remain the same, and we could expect the Earth to warm up by 1.6°C so as to once again arrive at thermal equilibrium.
The AGW hypothesis states that in the event of the Earth warming, changes in cloud cover will be such as to amplify the warming we calculate using Stefan's Law. Here is an extract from today's entry on Global Warming at Wikipedia.
The main positive feedback in the climate system is the water vapor feedback. The main negative feedback is radiative cooling through the Stefan–Boltzmann law, which increases as the fourth power of temperature.
As the Earth warms up, water evaporates more quickly from the oceans. Almost all water that evaporates must turn into clouds before it returns to Earth. Water condensing directly onto grass in the morning is an exception to this rule, but the vast majority of water vapor will return only as rain or snow, and so must first take the form of a cloud.
Clouds absorb long-wave radiation emitted by the Earth's surface, so the Earth cannot radiate its heat directly into space. Instead, the clouds radiate into space and warm the Earth with back radiation. Thus increasing cloud cover means less heat radiated into space for the same surface temperature. This is the positive feedback referred to by the AGW hypothesis. The AGW climate models predict that this positive feedback will amplify the minimum 1.5°C warming caused by CO2 to roughly 3°C. Some say 2°C and other say 4°C, but all agree that the actual warming will be greater than 1.5°C.
We see this positive feedback in our Circulating Cells simulation, version CC11, which simulates the formation of clouds as well as their absorption and emission of long-wave radiation. The graph below shows a close-up of the behavior of the simulation in the neighborhood of its equilibrium point for 350 W/m2 solar power.

The blue line shows how the power that penetrates to the surface of our simulated planet varies with increasing surface temperature. The orange line shows how the total power escaping from our simulated planet increases with surface temperature. These two lines cross at a, where temperature is 288 K and total escaping power is 290 W/m2. Our simulated atmosphere absorbs 50% of long-wave radiation, which is an adequate approximation of our atmosphere with its current concentration of CO2 (roughly 330 ppm).
The green line is the same as the orange line, but displaced down by 6.6 W/m2, which is the amount by which we calculated the total power escaping from the Earth will decrease if we double CO2 concentration (to roughly 660 ppm). Thus the green line tells us the total escaping power at the same temperature if we were to double the CO2 concentration. Point b on the green line is 288 K, and the total escaping power is 283.4 W/m2.

The purple line shows how the total escaping power will increase from b if we assume the cloud cover is constant and use only Stefan's Law to determine the heat radiated into space by the surface and atmosphere. The red line shows the solar power penetrating to the surface if we assume the cloud cover is constant. With constant cloud cover, the penetrating solar power does not change.
The purple and red lines meet at c, which is 289.6 K, or 1.6°C above the previous equilibrium point. Thus our simulation shows us that the warming due to CO2 doubling, if we ignore changes in cloud cover, will be 1.6°C, which is consistent with our previous calculation.
The green line, however, is the simulation's calculation of the total escaping power for increasing surface temperature. We see that the heat radiated into space does not increase as quickly as Stefan's Law would lead us to expect. And the reason for that is precisely the reason quoted by the AGW hypothesis: increasing cloud cover is slowing down the radiation of heat into space. The green line and the red line intersect at d, which is 290.7 K, or 2.7°C above our original equilibrium temperature. This is the new equilibrium temperature of the planet surface if we double CO2 concentration and we assume that there will be no change in the solar power penetrating to the surface while the cloud cover increases.

But the solar power penetrating to the surface must decrease as cloud cover increases. Clouds reflect sunlight. Thick clouds reflect 90% of solar power back into space. Even thin, high clouds reflect 10%. Increasing cloud cover will decrease the solar power penetrating to the surface. That is why our blue line slopes downwards. This is negative feedback, which acts against the positive feedback described by the AGW theory. The blue line shows how the solar power penetrating to the surface decreases as our cloud cover increases.
The blue line and the green line intersect at e, which is the equilibrium point we arrive at after doubling the CO2 concentration and considering both the positive feedback of back-radiation and the negative feedback of solar reflection. The temperature at e is 288.9 K, which is 0.9°C above our original equilibrium temperature.
Thus our simulation shows how the negative feedback generated by clouds dominates their positive feedback, and suggests that the actual warming of the Earth's surface due to a doubling of CO2 will be closer to 0.9°C than the 3°C predicted by the AGW hypothesis.
Labels:
Climate Models,
Global Surface,
Greenhouse Effect,
Water Vapor
Sunday, February 26, 2012
Thickening Clouds
As we describe in Solar Increase, we warmed up our simulated planet by increasing the incoming solar power by 10 W/m2 every two thousand hours of simulated time. Starting from an initial value of 100 W/m2, we increased the solar power to 1200 W/m2 over the course of three weeks of our own time, which corresponds to the passage of over a million hours of simulated time. During the course of the simulation, we recorded the state of the atmospheric array every twenty hours, and these recordings constitute our measurements of the simulated atmosphere during the course of our simulated experiment.
In our pervious post we observed that some properties of the atmosphere, such as penetrating power, fluctuated greatly from one measurement to the next. In order to reduce the influence of these fluctuations, we took the average of the last 500 hours of measurements at each value of incoming solar power, and so obtained a value for each property at each solar power. The graph below shows how some of these properties vary with solar power.
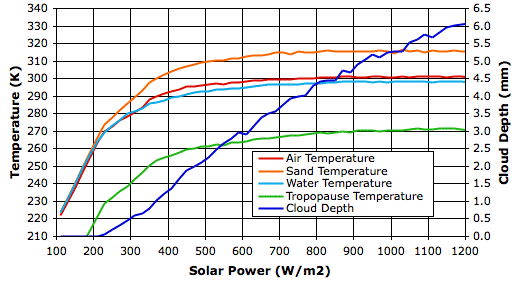
Surface temperature increases hardly at all from 800 W/m2 to 1200 W/m2, and yet cloud cover increases steadily. How can it be that cloud cover increases when the surface temperature, which drives evaporation, hardly increases at all?
In our simulated evaporation cycle, precipitation beings with the formation of snow in air below temperature Tf_droplets. We have this parameter set to 268 K, which is five degrees below the freezing point of water. When solar power reaches 800 W/m2, the average temperature of the tropopause has reached 268 K. Snow can form only in the colder clouds of the tropopause, and nowhere below the tropopause. Each time we increase the solar power, the surface temperature at first warms a little, but within a few hundred hours, this warming reaches the tropopause, where it further slows snow formation, and increases the cloud depth. With more sunlight being reflected back into space, the surface cools again until it is hardly warmer than it started. For solar powers greater than 800 W/m2, an increase of 100 W/m2 causes a substantial increase in cloud depth (roughly 0.5 mm), a slight increase in tropopause temperature (roughly 1 K), and an increase in surface temperature too small for us to detect (less than 0.3 K).
This profound suppression of warming by our simulation is not, however, a good representation of what would happen in the Earth's atmosphere. In our simulation, gas cells that contain clouds cannot rise above our top row of cells, so there is a limit to how much they can cool down. In the Earth's atmosphere, clouds can rise as far as they need to in order to cool down and produce snow rapidly. Thus our simulation is no longer realistic once its tropopause approaches the melting point of ice. We will therefore concentrate our attention upon the behavior of the simulation for solar powers less than 600 W/m2, for which our simulated tropopause is well below the temperature required for the rapid formation of snow.
In our pervious post we observed that some properties of the atmosphere, such as penetrating power, fluctuated greatly from one measurement to the next. In order to reduce the influence of these fluctuations, we took the average of the last 500 hours of measurements at each value of incoming solar power, and so obtained a value for each property at each solar power. The graph below shows how some of these properties vary with solar power.

Surface temperature increases hardly at all from 800 W/m2 to 1200 W/m2, and yet cloud cover increases steadily. How can it be that cloud cover increases when the surface temperature, which drives evaporation, hardly increases at all?
In our simulated evaporation cycle, precipitation beings with the formation of snow in air below temperature Tf_droplets. We have this parameter set to 268 K, which is five degrees below the freezing point of water. When solar power reaches 800 W/m2, the average temperature of the tropopause has reached 268 K. Snow can form only in the colder clouds of the tropopause, and nowhere below the tropopause. Each time we increase the solar power, the surface temperature at first warms a little, but within a few hundred hours, this warming reaches the tropopause, where it further slows snow formation, and increases the cloud depth. With more sunlight being reflected back into space, the surface cools again until it is hardly warmer than it started. For solar powers greater than 800 W/m2, an increase of 100 W/m2 causes a substantial increase in cloud depth (roughly 0.5 mm), a slight increase in tropopause temperature (roughly 1 K), and an increase in surface temperature too small for us to detect (less than 0.3 K).
This profound suppression of warming by our simulation is not, however, a good representation of what would happen in the Earth's atmosphere. In our simulation, gas cells that contain clouds cannot rise above our top row of cells, so there is a limit to how much they can cool down. In the Earth's atmosphere, clouds can rise as far as they need to in order to cool down and produce snow rapidly. Thus our simulation is no longer realistic once its tropopause approaches the melting point of ice. We will therefore concentrate our attention upon the behavior of the simulation for solar powers less than 600 W/m2, for which our simulated tropopause is well below the temperature required for the rapid formation of snow.
Labels:
Climate Models,
Greenhouse Effect,
Water Vapor
Thursday, February 23, 2012
Solar Increase Continued
Today we continue our previous post without any preamble. The graph below shows how our simulated atmosphere warms up as we increase the solar power from one hundred to twelve hundred Watts per square meter. The green line shows how we increased the solar power over the course of twelve simulated years. The blue line shows how the power penetrating to the surface varied with time. The red line is the average temperature of the air resting upon the surface of our simulated planet.
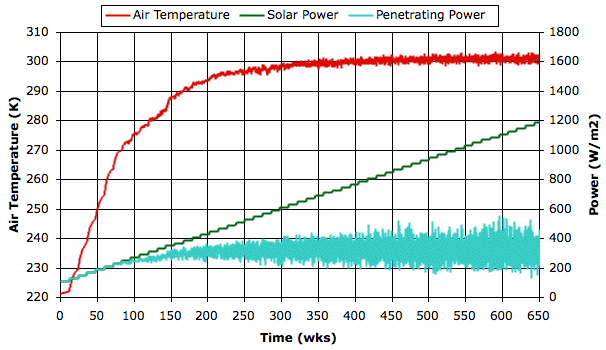
At first, when the sky is clear, the solar power and the penetrating power are equal. But when the solar power reaches 300 W/m2 clouds form and the penetrating power drops below the solar power. As solar power increases from 600 W/m2 to 1200 W/m2, fluctuations in the penetrating power double in their extent, but the average penetrating power appears to remain unchanged. The negative feedback generated by the evaporation cycle is so powerful that the surface air temperature increases by only a few degrees while we double the solar power.
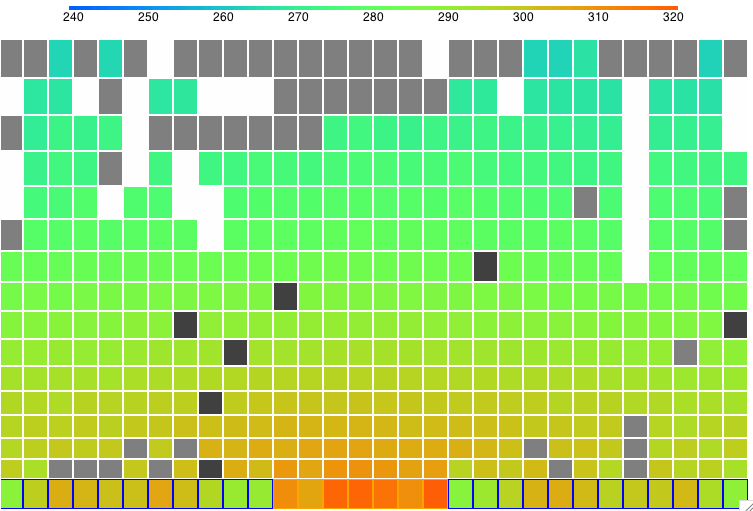
The following screen shot shows the state of the simulation after two thousand hours at 1210 W/m2. You can download this state as a text file SIC_1210W and load it into CC11 to watch the vigorous formation of clouds and descent of precipitation.
We now have the data we need to plot graphs of surface temperature and other properties of the simulation versus solar power.

At first, when the sky is clear, the solar power and the penetrating power are equal. But when the solar power reaches 300 W/m2 clouds form and the penetrating power drops below the solar power. As solar power increases from 600 W/m2 to 1200 W/m2, fluctuations in the penetrating power double in their extent, but the average penetrating power appears to remain unchanged. The negative feedback generated by the evaporation cycle is so powerful that the surface air temperature increases by only a few degrees while we double the solar power.
The following screen shot shows the state of the simulation after two thousand hours at 1210 W/m2. You can download this state as a text file SIC_1210W and load it into CC11 to watch the vigorous formation of clouds and descent of precipitation.

We now have the data we need to plot graphs of surface temperature and other properties of the simulation versus solar power.
Labels:
Climate Models,
Global Surface,
Greenhouse Effect,
Water Vapor
Monday, January 30, 2012
Lapse Rate
Our Radiating Clouds simulation uses 350 W/m2 for incoming solar power. We know from our Solar Heat calculation that this is the average power arriving at the Earth from the Sun. Our hope is that the lapse rate and surface temperature of our simulation will agree well with the actual lapse rate and surface temperature of the Earth's atmosphere. In the design of our simulation, however, we have made no effort to adjust its parameters to bring about such an agreement.
In the figure below, the blue graph shows temperature versus altitude for thermal equilibrium in the wet atmosphere of our Radiating Clouds simulation. We loaded the equilibrium state, RC_14000hr, into CC11 and instructed the simulation to print temperatures and altitudes.
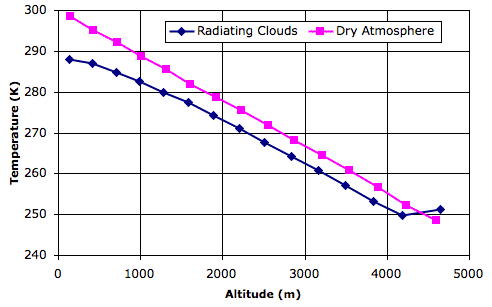
For comparison, we went back to Simulated Planet Surface and loaded the equilibrium state, Day_4, which arises with the same solar heat, but with the surface entirely made of sand. Thus the pink graph shows temperature versus altitude in a dry atmosphere.
The equilibrium state of the dry atmosphere, which looks like this, shows a linear drop in temperature with altitude. According to our calculations, this slope should be −g/Cp, where g is gravity and Cp is the specific heat capacity of the dry gas. Our simulation uses 10 N/kg for gravity and 1003 J/K for Cp, so the slope of the pink graph should be close to −0.010 K/m. Its actual slope is −0.011 K/m.
The equilibrium state of the wet atmosphere, which looks like this, shows a linear drop in temperature only between altitudes 2 km and 4 km. Near the surface of the planet, the heat liberated by condensing water vapor into rising air reduces the amount by which air cools as it rises. In the tropopause, radiation from the planet surface is absorbed by the tropopause clouds, causing them to be warmer than the air immediately below. In the linear region of the graph, the slope is −0.011 K/m, but if we simply divide the total drop in temperature by the altitude of the tropopause, we obtain a net slope of −0.008 K/m.
Looking at graphs such as these, it appears that the Earth's atmosphere has a lapse rate of around −0.0065 K/m. Our simulation does not come up with this lapse rate exactly, but we see that the introduction of water does cause a substantial reduction in the net lapse rate, and with this we are well-satisfied.
The average temperature of the surface of the Earth is around 14°C, or 287 K. This is the average temperature of the air just above the planet surface, not the temperature of the surface itself. The thermometers we use to measure the surface temperature are several meters above the ground. When we use dry air and a sandy surface in our simulation, the average temperature of the surface air is 298 K. But when we add radiating clouds, rain, and snow, the average temperature drops to 288 K, or 15°C. We are well-satisfied with this agreement also.
In the figure below, the blue graph shows temperature versus altitude for thermal equilibrium in the wet atmosphere of our Radiating Clouds simulation. We loaded the equilibrium state, RC_14000hr, into CC11 and instructed the simulation to print temperatures and altitudes.

For comparison, we went back to Simulated Planet Surface and loaded the equilibrium state, Day_4, which arises with the same solar heat, but with the surface entirely made of sand. Thus the pink graph shows temperature versus altitude in a dry atmosphere.
The equilibrium state of the dry atmosphere, which looks like this, shows a linear drop in temperature with altitude. According to our calculations, this slope should be −g/Cp, where g is gravity and Cp is the specific heat capacity of the dry gas. Our simulation uses 10 N/kg for gravity and 1003 J/K for Cp, so the slope of the pink graph should be close to −0.010 K/m. Its actual slope is −0.011 K/m.
The equilibrium state of the wet atmosphere, which looks like this, shows a linear drop in temperature only between altitudes 2 km and 4 km. Near the surface of the planet, the heat liberated by condensing water vapor into rising air reduces the amount by which air cools as it rises. In the tropopause, radiation from the planet surface is absorbed by the tropopause clouds, causing them to be warmer than the air immediately below. In the linear region of the graph, the slope is −0.011 K/m, but if we simply divide the total drop in temperature by the altitude of the tropopause, we obtain a net slope of −0.008 K/m.
Looking at graphs such as these, it appears that the Earth's atmosphere has a lapse rate of around −0.0065 K/m. Our simulation does not come up with this lapse rate exactly, but we see that the introduction of water does cause a substantial reduction in the net lapse rate, and with this we are well-satisfied.
The average temperature of the surface of the Earth is around 14°C, or 287 K. This is the average temperature of the air just above the planet surface, not the temperature of the surface itself. The thermometers we use to measure the surface temperature are several meters above the ground. When we use dry air and a sandy surface in our simulation, the average temperature of the surface air is 298 K. But when we add radiating clouds, rain, and snow, the average temperature drops to 288 K, or 15°C. We are well-satisfied with this agreement also.
Labels:
Climate Models,
Greenhouse Effect,
Water Vapor
Sunday, January 22, 2012
Thermal Equillibrium
An object is in thermal equilibrium when the amount of heat entering the object is equal to the amount of heat leaving it. In the case of a planetary system, consisting of a surface and atmosphere, the system will be in thermal equilibrium when the heat arriving from the Sun is, on average, equal to the heat the system radiates into space. The planetary system of our Circulating Cells program will be in thermal equilibrium when the short-wave radiation penetrating to the planet surface from the Sun is balanced by the long-wave radiation escaping into space from its surface, atmospheric gas, clouds, rain, and snow.
When our simulated system converges upon a state where the temperature of its surface and of its atmospheric layers fluctuates around some average value, our hope is that the heat penetrating to the surface from the Sun will be equal, on average, to the heat the system radiates into space. The heat penetrating from the Sun is, of course, the incoming short-wave radiation that is not reflected back into space by our simulated clouds. The heat radiated into space is the upwelling radiation at the top of our simulated atmosphere. We refer to the top of the atmosphere at its tropopause, and to the heat radiated into space as the total escaping power.
We instructed CC11 to print out the average penetrating power from the Sun and the total escaping power from the tropopause every ten hours, and plotted these values for the fourteen thousand hours of the experiment we performed in our previous post. For the final ten thousand hours, the temperature of the surface and the atmospheric layers fluctuate by ±1°C around their average values, as you can see here.

Over the final ten thousand hours, the average penetrating power is 290.0 W/m2 and the average escaping power is 289.6 W/m2. Given the size of the fluctuations in both quantities, and the errors introduced by certain simplifications in our simulation's calculations, we are well-satisfied with this agreement. Our simulation converges upon an equilibrium state that is also a state of thermal equilibrium.
When our simulated system converges upon a state where the temperature of its surface and of its atmospheric layers fluctuates around some average value, our hope is that the heat penetrating to the surface from the Sun will be equal, on average, to the heat the system radiates into space. The heat penetrating from the Sun is, of course, the incoming short-wave radiation that is not reflected back into space by our simulated clouds. The heat radiated into space is the upwelling radiation at the top of our simulated atmosphere. We refer to the top of the atmosphere at its tropopause, and to the heat radiated into space as the total escaping power.
We instructed CC11 to print out the average penetrating power from the Sun and the total escaping power from the tropopause every ten hours, and plotted these values for the fourteen thousand hours of the experiment we performed in our previous post. For the final ten thousand hours, the temperature of the surface and the atmospheric layers fluctuate by ±1°C around their average values, as you can see here.

Over the final ten thousand hours, the average penetrating power is 290.0 W/m2 and the average escaping power is 289.6 W/m2. Given the size of the fluctuations in both quantities, and the errors introduced by certain simplifications in our simulation's calculations, we are well-satisfied with this agreement. Our simulation converges upon an equilibrium state that is also a state of thermal equilibrium.
Wednesday, January 18, 2012
Radiating Clouds
The latest version of Circulating Cells implements the upwelling and downwelling radiation calculations we described in Up and Down Radiation. To run the program, download CC11 and follow the instructions at the top of the code. Clouds absorb and emit long-wave radiation as if they were black bodies. We now set the transparency fraction of our atmospheric gas to 0.5, so that it will be transparent to half the wavelengths in the long-wave spectrum and opaque otherwise. The planet surface can radiate heat directly into space at these transparent wavelengths, as it did in Simulated Planet Surface. But now we have clouds doing the same thing, while at the same time reflecting sunlight back into space.
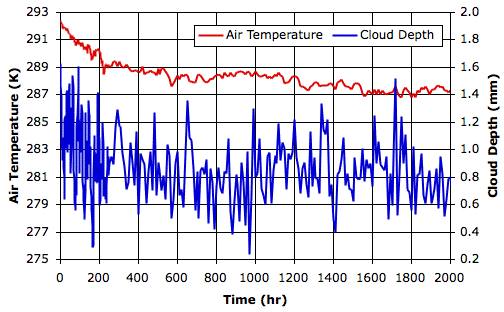
We begin our simulation with the final state of Simulated Rain, which you will find in SR_1200hr. The initial surface air temperature is 292 K, and cloud depth is 1.5 mm. The following graph shows how air temperature and cloud depth vary in the first two thousand hours.
The following graph shows the first fourteen thousand hours. You will find the final state of the array in RC_14000hr. The average surface air temperature over the final ten thousand hours is 288 K, and the average cloud depth is 0.8 mm.
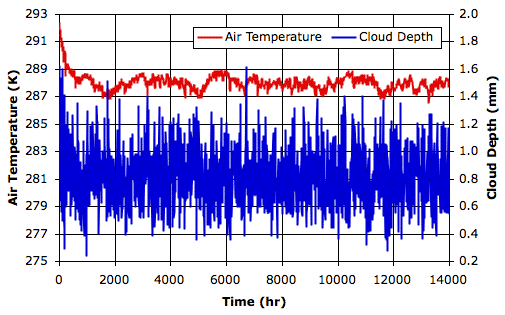
During the course of these fourteen thousand hours, the distribution of clouds in the atmosphere varies greatly. Sometimes there is a layer of clouds just above the surface of the sea. At other times there are clouds along much of the tropopause. For a view of the final state of the simulation, see here.
As we have discussed many times before, the absorption of long-wave radiation by the atmosphere gives rise to the greenhouse effect. The more opaque the atmosphere, the more heat must be radiated into space by the tropopause instead of the planet surface. In order to radiate more heat, the tropopause must be warmer. If the tropopause is warmer, the planet surface must be warmer too, in order to motivate convection to carry heat to the tropopause. When we change our atmospheric gas from 0% to 50% transparency, we expect the surface temperature drop. And indeed it does: by 4°C.
This cooling of 4°C is, however, far less than the cooling of 31°C we observed when we increased the transparency of our gas from 0% to 50% in the absence of simulated clouds. As we have already discussed, clouds and rain greatly reduce the sensitivity of surface temperature to changes in solar power. Now we find that they also greatly reduce the sensitivity of surface temperature to changes in the transparency of the atmospheric gas.
We begin our simulation with the final state of Simulated Rain, which you will find in SR_1200hr. The initial surface air temperature is 292 K, and cloud depth is 1.5 mm. The following graph shows how air temperature and cloud depth vary in the first two thousand hours.

The following graph shows the first fourteen thousand hours. You will find the final state of the array in RC_14000hr. The average surface air temperature over the final ten thousand hours is 288 K, and the average cloud depth is 0.8 mm.

During the course of these fourteen thousand hours, the distribution of clouds in the atmosphere varies greatly. Sometimes there is a layer of clouds just above the surface of the sea. At other times there are clouds along much of the tropopause. For a view of the final state of the simulation, see here.
As we have discussed many times before, the absorption of long-wave radiation by the atmosphere gives rise to the greenhouse effect. The more opaque the atmosphere, the more heat must be radiated into space by the tropopause instead of the planet surface. In order to radiate more heat, the tropopause must be warmer. If the tropopause is warmer, the planet surface must be warmer too, in order to motivate convection to carry heat to the tropopause. When we change our atmospheric gas from 0% to 50% transparency, we expect the surface temperature drop. And indeed it does: by 4°C.
This cooling of 4°C is, however, far less than the cooling of 31°C we observed when we increased the transparency of our gas from 0% to 50% in the absence of simulated clouds. As we have already discussed, clouds and rain greatly reduce the sensitivity of surface temperature to changes in solar power. Now we find that they also greatly reduce the sensitivity of surface temperature to changes in the transparency of the atmospheric gas.
Labels:
Climate Models,
Greenhouse Effect,
Water Vapor
Friday, January 13, 2012
Up and Down Radiation
We are going to add to our Circulating Cells simulation the absorption and emission of long-wave radiation by clouds. As we showed earlier, a liquid water depth of 100 μm absorbs over 99% of all long-wave radiation. Rain contains liquid water also, and ice is a good absorber of long-wave radiation too. We will add the equivalent depth of snow, rain, and cloud droplets for each cell, and so obtain the depth of water within the cell that acts to absorb long-wave radiation.
We note that the same addition of rain, snow, and cloud droplets does not apply to the transmission of short-wave radiation. Water is transparent to short-wave radiation, and clouds reflect it by refracting it through millions of microscopic droplets. But rain and snow contain thousands of times fewer drops and crystals for a given depth of water, so they are thousands of times less effective at refracting sunlight.
For simplicity, we will assume the water in a cell is either transparent or opaque to long-wave radiation, but not in-between. If the combined concentration of rain, snow, and cloud droplets in a cell is greater than wc_opaque, we will assume the entire gas cell is opaque to long-wave radiation. Otherwise, the cell will absorb long-wave radiation as if it were dry, as determined by our transparency_fraction. With our 300-kg cells, a concentration of 0.33 g/kg corresponds to 100 μm of water.
Now we are faced with the possibility of multiple layers of cloud, snow, and rain, all absorbing and emitting long-wave radiation in all directions. The first simplification we make is to assume each gas cell radiates only vertically upwards and downwards. Because our columns of cells are much the same as one another on average, the net effect of this simplification will be small. Even with this simplification, we see that a cloud can absorb radiation from a cloud below, and emit radiation back to that same cloud below, and upwards to a third cloud.
We will calculate the effect of long-wave radiation in the following way. We start at the surface and allow it to radiate as a black body. We allow this upward radiation to enter the first gas cell. We calculate how much is absorbed by the cell and how much keeps going. We calculate how much power the gas cell itself radiates upwards. We add this to the existing upward radiation. We move on to the cell above, and so on, until we get to the tropopause. At the tropopause, we assume the atmosphere above is transparent to long-wave radiation, so all upward-going radiation passes out into space.
We repeat the same process, going down. We start with the tropopause gas cell in each column and move down cell by cell until we arrive at the bottom, at which point all the downward-going radiation is absorbed by the surface. We first considered this kind of downward-going long-wave radiation in our Back Radiation post. It is distinct from the solar radiation that penetrates the atmospheric clouds because it is radiation emitted by the clouds, rain, snow, and atmospheric gas themselves.
In any cell, the long-wave radiation going up is the upwelling radiation and the long-wave radiation going down is the downwelling radiation. At the tropopause, the upwelling radiation is the heat leaving our planetary system. It is our total escaping power. When our simulation converges to equilibrium, we should find that the average solar power penetrating to the surface is equal to the average total escaping power.
We note that the same addition of rain, snow, and cloud droplets does not apply to the transmission of short-wave radiation. Water is transparent to short-wave radiation, and clouds reflect it by refracting it through millions of microscopic droplets. But rain and snow contain thousands of times fewer drops and crystals for a given depth of water, so they are thousands of times less effective at refracting sunlight.
For simplicity, we will assume the water in a cell is either transparent or opaque to long-wave radiation, but not in-between. If the combined concentration of rain, snow, and cloud droplets in a cell is greater than wc_opaque, we will assume the entire gas cell is opaque to long-wave radiation. Otherwise, the cell will absorb long-wave radiation as if it were dry, as determined by our transparency_fraction. With our 300-kg cells, a concentration of 0.33 g/kg corresponds to 100 μm of water.
Now we are faced with the possibility of multiple layers of cloud, snow, and rain, all absorbing and emitting long-wave radiation in all directions. The first simplification we make is to assume each gas cell radiates only vertically upwards and downwards. Because our columns of cells are much the same as one another on average, the net effect of this simplification will be small. Even with this simplification, we see that a cloud can absorb radiation from a cloud below, and emit radiation back to that same cloud below, and upwards to a third cloud.
We will calculate the effect of long-wave radiation in the following way. We start at the surface and allow it to radiate as a black body. We allow this upward radiation to enter the first gas cell. We calculate how much is absorbed by the cell and how much keeps going. We calculate how much power the gas cell itself radiates upwards. We add this to the existing upward radiation. We move on to the cell above, and so on, until we get to the tropopause. At the tropopause, we assume the atmosphere above is transparent to long-wave radiation, so all upward-going radiation passes out into space.
We repeat the same process, going down. We start with the tropopause gas cell in each column and move down cell by cell until we arrive at the bottom, at which point all the downward-going radiation is absorbed by the surface. We first considered this kind of downward-going long-wave radiation in our Back Radiation post. It is distinct from the solar radiation that penetrates the atmospheric clouds because it is radiation emitted by the clouds, rain, snow, and atmospheric gas themselves.
In any cell, the long-wave radiation going up is the upwelling radiation and the long-wave radiation going down is the downwelling radiation. At the tropopause, the upwelling radiation is the heat leaving our planetary system. It is our total escaping power. When our simulation converges to equilibrium, we should find that the average solar power penetrating to the surface is equal to the average total escaping power.
Labels:
Climate Models,
Greenhouse Effect,
Water Vapor
Monday, November 28, 2011
Less Reflection
With 350 W/m2 arriving from the Sun, 75% of the surface covered by water, clouds sinking at 300 mm/s, and each 3 mm of cloud reflecting 63% of sunlight, our CC9 simulation converges upon a surface air temperature of −12°C. When we increase the Sun's power to 400 W/m2, the temperature rises by a mere 0.5°C. Our simulated planet is kept cold by thick clouds that reflect the Sun's light back into space. Ice crystals drift down from the sky in some places, while elsewhere water evaporates from the frozen seas.
The surface of the Earth is at an average temperature well above the freezing point of water, and the Earth's sky is frequently clear of clouds. Our simulated sky never clears, and the surface is frozen. It never rains in our simulation, nor do our simulated clouds emit or absorb radiation. Perhaps these two omissions are responsible for our permanent clouds and frozen seas. Before we attempt to rectify them, however, let us consider the effect of decreasing the reflecting power of our simulated clouds.
We increased Lc_water from 3.0 mm to 6.0 mm, so that it now takes 6.0 mm of cloud water to reflect 63% of the Sun's light. With the reflecting power divided in half, we ran our simulation for eight thousand hours from the starting point CS_0hr. You will find the final state in LR_8000hr.

Compared to before, we now have more clouds in the sky. The following graph shows how cloud depth and surface air temperature vary with time.
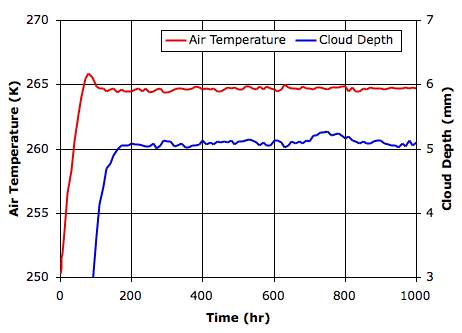
Compared to before, we see the atmosphere reaches equilibrium in on third the time. The new temperature is higher and the cloud cover is thicker. The following table compares the state of the atmosphere for both types of clouds.

Our seas are now at −3°C. If they contain salt, they will not freeze. The air a few meters above our sandy island will be just below freezing. Our simulated world is still much colder than the Earth, and nobody standing on the island would ever see the Sun. We are, however, gratified to find that our simulation remains stable with such a large drop in cloud reflectance.
The surface of the Earth is at an average temperature well above the freezing point of water, and the Earth's sky is frequently clear of clouds. Our simulated sky never clears, and the surface is frozen. It never rains in our simulation, nor do our simulated clouds emit or absorb radiation. Perhaps these two omissions are responsible for our permanent clouds and frozen seas. Before we attempt to rectify them, however, let us consider the effect of decreasing the reflecting power of our simulated clouds.
We increased Lc_water from 3.0 mm to 6.0 mm, so that it now takes 6.0 mm of cloud water to reflect 63% of the Sun's light. With the reflecting power divided in half, we ran our simulation for eight thousand hours from the starting point CS_0hr. You will find the final state in LR_8000hr.

Compared to before, we now have more clouds in the sky. The following graph shows how cloud depth and surface air temperature vary with time.

Compared to before, we see the atmosphere reaches equilibrium in on third the time. The new temperature is higher and the cloud cover is thicker. The following table compares the state of the atmosphere for both types of clouds.

Our seas are now at −3°C. If they contain salt, they will not freeze. The air a few meters above our sandy island will be just below freezing. Our simulated world is still much colder than the Earth, and nobody standing on the island would ever see the Sun. We are, however, gratified to find that our simulation remains stable with such a large drop in cloud reflectance.
Labels:
Climate Models,
Global Surface,
Greenhouse Effect,
Water Vapor
Wednesday, October 19, 2011
Cold Start
In our previous post, we presented our simulation of clouds without rain. We started the atmospheric gas, the sandy island, and the watery sea, at a uniform 280 K (7°C). Water evaporated from the sea. The sand heated up in the sun. Hot air rose above the island and sucked moist air in from the sea. Clouds formed above the island, spread through the atmosphere, reflected the heat of the sun, and the world froze.
What if we start with a frozen world and a dry atmosphere? In our simulation of evaporation rate, no water will evaporate from a sea at 250 K (−23°C), so no clouds will form. We ran CC9, starting with the CS_0hr array, to find out what would happen. Our starting point is a uniform 250 K with no water vapor. We run with 350 W/m2 continuous heat from the Sun.
After 20 hrs, the sandy island has warmed to 276 K (3°C). At 30 hrs, the average cloud depth is 0.03 mm, which is so thin that we don't bother plotting the clouds as white cells. But at 40 hrs we start to see the first thin clouds, and the average power arriving from the Sun drops to 335 W/m2. At 50 hrs, the island reaches 283 K (10°C). From here on, it cools. At 100 hrs, the average cloud depth is 3.5 mm and only 120 W/m2 is arriving from the Sun. The sea reaches 267 K (−6°C), which is the warmest it will ever get. By 200 hrs, cloud depth is 7.2 mm and power arriving from the Sun is only 40 W/m2, as recored in CS_200hr.
We can see where the simulation is going to end up: a world kept frozen by immortal clouds. Regardless of our starting point, immortal clouds reflect the Sun's heat and cause the world to freeze.
What if we start with a frozen world and a dry atmosphere? In our simulation of evaporation rate, no water will evaporate from a sea at 250 K (−23°C), so no clouds will form. We ran CC9, starting with the CS_0hr array, to find out what would happen. Our starting point is a uniform 250 K with no water vapor. We run with 350 W/m2 continuous heat from the Sun.
After 20 hrs, the sandy island has warmed to 276 K (3°C). At 30 hrs, the average cloud depth is 0.03 mm, which is so thin that we don't bother plotting the clouds as white cells. But at 40 hrs we start to see the first thin clouds, and the average power arriving from the Sun drops to 335 W/m2. At 50 hrs, the island reaches 283 K (10°C). From here on, it cools. At 100 hrs, the average cloud depth is 3.5 mm and only 120 W/m2 is arriving from the Sun. The sea reaches 267 K (−6°C), which is the warmest it will ever get. By 200 hrs, cloud depth is 7.2 mm and power arriving from the Sun is only 40 W/m2, as recored in CS_200hr.
We can see where the simulation is going to end up: a world kept frozen by immortal clouds. Regardless of our starting point, immortal clouds reflect the Sun's heat and cause the world to freeze.
Labels:
Climate Models,
Greenhouse Effect,
Water Vapor,
Weather
Tuesday, October 4, 2011
Clouds Without Rain
Today we present Circulating Cells Version 9 (CC9), and we use it to find out what would happen if we had clouds without rain. The simulation implements the following features of clouds.
(1) Evaporation from surface water, as in Evaporation Rate.
(2) Condensation in rising air, as in Condensation Point and Condensation Rate.
(3) Cooling and warming by latent heat of evaporation, as in Latent Heat.
(4) Reflection of incoming sunlight, as in Simulated Clouds, Part I.
The simulation does not yet implement the following features of clouds.
(5) Absorption and emission of long-wave radiation, as in Simulated Clouds, Part II.
(6) Cooling and warming by latent heat of fusion, as in Latent Heat.
(7) Rain and snow.
If we set the simulated atmosphere's transparency fraction to 0.0, we make the atmospheric gas opaque to long-wave radiation. No radiation escapes into space from the surface blocks nor from the rows of gas cells below the top row, regardless of the distribution of clouds within the atmosphere. The top row of cells, which is our simulated tropopause, does all the radiating of heat into space. Although this opaque atmosphere is not realistic, it does mask the fact that our clouds do not in themselves absorb or emit long-wave radiation, allowing us to proceed with a simulation that is at least self-consistent. Thus the copy of CC9 that you can download today has the transparency fraction set to 0.0 by default.
We ignore the warming of rising air by freezing water droplets, and the cooling of falling air by melting ice crystals. We will add ice crystals to our simulation later. For now, we trust that the error caused by our omission is not so great as to overturn the observations we make today.
By ignoring rain and snow, we are ignoring a feature of clouds that is so important to our climate that our simulation produces an entirely fantastic result. To watch the simulation in action, download CC9 and follow the instructions at the top of the code to run the program on your computer. Get the CWR_0hr array file and load it with the Load button. You will see an atmosphere at a uniform 280 K, and down at the bottom, an island of sand in a sea of water, also at 280 K. Press Run and the simulation will begin. The CC9 code runs in "Day" mode by default, with 350 W/m2 arriving continuously from the sun.
Without rain and snow, any and all moisture that enters the atmosphere at the beginning of the simulation remains in the atmosphere for as long as the simulation runs. There is no means by which moisture can return to the surface of the planet. So long as the lower atmosphere is warm enough to absorb water vapor, however, the clouds can appear and disappear. The moisture they contain can either take the form of water vapor, as it will when the surrounding gas is warm, or it can take the form of water droplets, as it will when the surrounding gas is cold.
We represent clouds in our simulation with cells that are shaded white to gray. The thinnest clouds are white and the thickest are black. A cloud with 1 mm of water is white. A cloud with 12 mm of water is black. After 30 hours, the first white clouds appear over the island, the result of moist air from the sea being heated by the island and rising towards the tropopause. When it rises, it cools, and water vapor condenses to form the first clouds.
At 40 hours, the island reaches its peak temperature of around 311 K. After that, the clouds become more numerous. They reflect the Sun's light back into space. The surface begins to cool. After 150 hrs we end up with the following display.
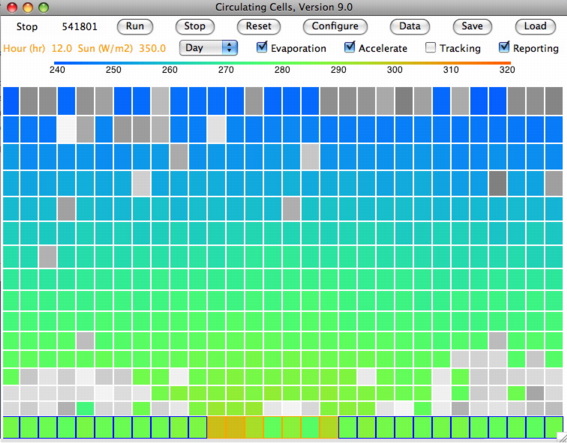
There is fog over the sea and part of the island. There are thick clouds up in the tropopause. The average heat arriving at the surface from the Sun has dropped from 350 W/m2 to only 50 W/m2. The following graph shows how the atmosphere cools in the first 500 hrs.
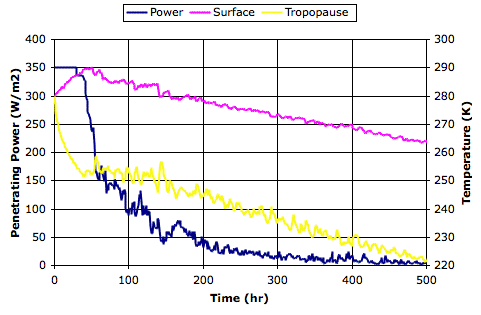
Let us refer to the combined thickness of the clouds above a surface block as is its cloud cover. In our simulation, each 3 mm of cloud cover reflects 63% of incoming sunlight. If we press the Data button, a text window opens and here we will see a line of numbers printed every hour of simulation time. The first number is the time in hours, the second is the average cloud cover in millimeters. The third number is the average sunlight power penetrating to the surface through the cloud cover in Watt per square meter. After that we have four temperatures in Kelvin: average sand temperature, average water temperature, average surface gas temperature, and average tropopause temperature. We used these printed lines to obtain the data for the plot above.
After 3000 hrs, the tropopause has dropped to 158 K (−115°C) and the surface air is at 184 K (−85°C). The average cloud cover is 25 mm. Only 0.7 W/m2 arrives at the surface. You can see this for yourself by loading CWR_3000hr into the simulation. Even at 158 K, the tropopause is still radiating 35 W/m2, which is far more than the 0.7 W/m2 reaching the planet surface. The tropopause will keep cooling until it reaches 60 K, at which point it will be radiating 0.7 W/m2. Of course, at that point, nitrogen will condense into liquid.
If clouds remained aloft in the atmosphere indefinitely, the Earth would freeze. But in reality, clouds are forever falling towards the ground. They are made of droplets and crystals that are heavier than air. Rain and snow are what stop clouds from turning the Earth into a planet of frozen seas.
(1) Evaporation from surface water, as in Evaporation Rate.
(2) Condensation in rising air, as in Condensation Point and Condensation Rate.
(3) Cooling and warming by latent heat of evaporation, as in Latent Heat.
(4) Reflection of incoming sunlight, as in Simulated Clouds, Part I.
The simulation does not yet implement the following features of clouds.
(5) Absorption and emission of long-wave radiation, as in Simulated Clouds, Part II.
(6) Cooling and warming by latent heat of fusion, as in Latent Heat.
(7) Rain and snow.
If we set the simulated atmosphere's transparency fraction to 0.0, we make the atmospheric gas opaque to long-wave radiation. No radiation escapes into space from the surface blocks nor from the rows of gas cells below the top row, regardless of the distribution of clouds within the atmosphere. The top row of cells, which is our simulated tropopause, does all the radiating of heat into space. Although this opaque atmosphere is not realistic, it does mask the fact that our clouds do not in themselves absorb or emit long-wave radiation, allowing us to proceed with a simulation that is at least self-consistent. Thus the copy of CC9 that you can download today has the transparency fraction set to 0.0 by default.
We ignore the warming of rising air by freezing water droplets, and the cooling of falling air by melting ice crystals. We will add ice crystals to our simulation later. For now, we trust that the error caused by our omission is not so great as to overturn the observations we make today.
By ignoring rain and snow, we are ignoring a feature of clouds that is so important to our climate that our simulation produces an entirely fantastic result. To watch the simulation in action, download CC9 and follow the instructions at the top of the code to run the program on your computer. Get the CWR_0hr array file and load it with the Load button. You will see an atmosphere at a uniform 280 K, and down at the bottom, an island of sand in a sea of water, also at 280 K. Press Run and the simulation will begin. The CC9 code runs in "Day" mode by default, with 350 W/m2 arriving continuously from the sun.
Without rain and snow, any and all moisture that enters the atmosphere at the beginning of the simulation remains in the atmosphere for as long as the simulation runs. There is no means by which moisture can return to the surface of the planet. So long as the lower atmosphere is warm enough to absorb water vapor, however, the clouds can appear and disappear. The moisture they contain can either take the form of water vapor, as it will when the surrounding gas is warm, or it can take the form of water droplets, as it will when the surrounding gas is cold.
We represent clouds in our simulation with cells that are shaded white to gray. The thinnest clouds are white and the thickest are black. A cloud with 1 mm of water is white. A cloud with 12 mm of water is black. After 30 hours, the first white clouds appear over the island, the result of moist air from the sea being heated by the island and rising towards the tropopause. When it rises, it cools, and water vapor condenses to form the first clouds.
At 40 hours, the island reaches its peak temperature of around 311 K. After that, the clouds become more numerous. They reflect the Sun's light back into space. The surface begins to cool. After 150 hrs we end up with the following display.

There is fog over the sea and part of the island. There are thick clouds up in the tropopause. The average heat arriving at the surface from the Sun has dropped from 350 W/m2 to only 50 W/m2. The following graph shows how the atmosphere cools in the first 500 hrs.

Let us refer to the combined thickness of the clouds above a surface block as is its cloud cover. In our simulation, each 3 mm of cloud cover reflects 63% of incoming sunlight. If we press the Data button, a text window opens and here we will see a line of numbers printed every hour of simulation time. The first number is the time in hours, the second is the average cloud cover in millimeters. The third number is the average sunlight power penetrating to the surface through the cloud cover in Watt per square meter. After that we have four temperatures in Kelvin: average sand temperature, average water temperature, average surface gas temperature, and average tropopause temperature. We used these printed lines to obtain the data for the plot above.
After 3000 hrs, the tropopause has dropped to 158 K (−115°C) and the surface air is at 184 K (−85°C). The average cloud cover is 25 mm. Only 0.7 W/m2 arrives at the surface. You can see this for yourself by loading CWR_3000hr into the simulation. Even at 158 K, the tropopause is still radiating 35 W/m2, which is far more than the 0.7 W/m2 reaching the planet surface. The tropopause will keep cooling until it reaches 60 K, at which point it will be radiating 0.7 W/m2. Of course, at that point, nitrogen will condense into liquid.
If clouds remained aloft in the atmosphere indefinitely, the Earth would freeze. But in reality, clouds are forever falling towards the ground. They are made of droplets and crystals that are heavier than air. Rain and snow are what stop clouds from turning the Earth into a planet of frozen seas.
Labels:
Climate Models,
Greenhouse Effect,
Water Vapor,
Weather
Tuesday, September 27, 2011
Summary to Date
Once we were satisfied that our simulation handled convection properly, that we could relate the program iterations to the passage of time, and that all the heat entering the simulated system was accounted for by radiation from the top, we added blocks of either water or sand beneath the bottom gas cells, so as to simulate the planet surface. In Back Radiation we showed how the heat capacity and radiation produced by a semi-transparent atmosphere keeps the planet surface warm at night. In Island Inversion we see the surface of an island heating up ten times more than the surrounding ocean, while at night a layer of air a few hundred meters above the island is warmer, rather than cooler, than the air resting upon the island. Thus we see our simulation is consistent with our observations of surface cooling, including even temperature inversion.
Well-satisfied with our simulation of a dry atmosphere, we now turn to the simulation of a wet atmosphere, in which evaporation will cool the ocean and lead to the formation of clouds. To simulate cloud formation, we must have equations for the rate of evaporation from a water surface, the rate at which water vapor will condense out of rising air, the rate at which it will evaporate again in falling air, the cooling effect of evaporation upon the water surface, the warming effect of condensation upon the rising air, the amount of sunlight that will be reflected by existing clouds, and the amount of long-wave radiation that these same clouds will absorb and radiate. We obtained these relations in a series of posts Evaporation Rate to Consensation Rate. We have yet to consider the downward drift of water droplets that leads to their combining together and forming rain. But after so many posts of mathematics and empirical relations, we thought it was time to get back to the simulation, and so we will start our simulation of clouds without allowing rain, and perhaps we will see how important rain is for our climate.
We are running CC9 right now, and will present it later this week, once I have made a reasonable effort to eliminate errors from my implementation of evaporation, condensation, and reflection. The clouds are going round right now, as gray-shaded cells, and the effect is entertaining. Ultimately, you may recall, our objective is to see how a change in the transparency of the dry atmosphere affects the surface temperature of the planet, so that we can determine the effect of CO2 doubling within a system dominated by the effect of cloud formation and rain.
Well-satisfied with our simulation of a dry atmosphere, we now turn to the simulation of a wet atmosphere, in which evaporation will cool the ocean and lead to the formation of clouds. To simulate cloud formation, we must have equations for the rate of evaporation from a water surface, the rate at which water vapor will condense out of rising air, the rate at which it will evaporate again in falling air, the cooling effect of evaporation upon the water surface, the warming effect of condensation upon the rising air, the amount of sunlight that will be reflected by existing clouds, and the amount of long-wave radiation that these same clouds will absorb and radiate. We obtained these relations in a series of posts Evaporation Rate to Consensation Rate. We have yet to consider the downward drift of water droplets that leads to their combining together and forming rain. But after so many posts of mathematics and empirical relations, we thought it was time to get back to the simulation, and so we will start our simulation of clouds without allowing rain, and perhaps we will see how important rain is for our climate.
We are running CC9 right now, and will present it later this week, once I have made a reasonable effort to eliminate errors from my implementation of evaporation, condensation, and reflection. The clouds are going round right now, as gray-shaded cells, and the effect is entertaining. Ultimately, you may recall, our objective is to see how a change in the transparency of the dry atmosphere affects the surface temperature of the planet, so that we can determine the effect of CO2 doubling within a system dominated by the effect of cloud formation and rain.
Labels:
Climate Models,
CO2,
Greenhouse Effect,
Water Vapor,
Weather
Wednesday, August 3, 2011
Back Radiation
In our Rotating Greenhouse post, we use CC5 to simulate the alternation between day and night by varying the Solar power delivered to the gas cells resting upon our simulated planet surface. But we did not simulate the surface itself, nor did we distinguish between the temperature of the surface gas and the radiating temperature of the planet surface: we used the same temperature. But the CC8 program we introduced in our previous post does simulate the planet surface, so we can see how the temperature of the sand itself varies with day and night.
During the day, most of the heat passing into the surface gas cells does so by convection. But convection occurs only when the gas above is cooler than the surface below. In our discussion of atmospheric inversion we saw how the ground can be colder than the surface air at night, which can lead to a pocket of cold air sitting near the ground, with warmer air up above. In our simulation, we set the convection transfer to zero when the surface is colder than the surface gas.
At night, therefore, a sandy surface will radiate its heat into space, and receive no warmth from the sun. But it will receive warmth from the atmosphere, in the form of the back-radiation we described in our previous post. During the day, we found that our surface gas was radiating 226 W/m2 down to the surface. This radiation will slow the cooling of the surface at night.
We ran our simulation with Cycle heating on a sandy planet surface, 700 W/m2 solar heat during the day, daylight fraction set to 0.50, convection coefficient 20 W/m2, and transparency fraction 0.50. You will find the equilibrium state of the cell array at midnight stored in a text file here. The following graph shows the average temperature of the sand blocks, the surface gas cells, and the tropopause gas cells during two complete day-night cycles. We plot the deviation of each temperature from its average value during the cycles, which is why we call the plots "anomalies".
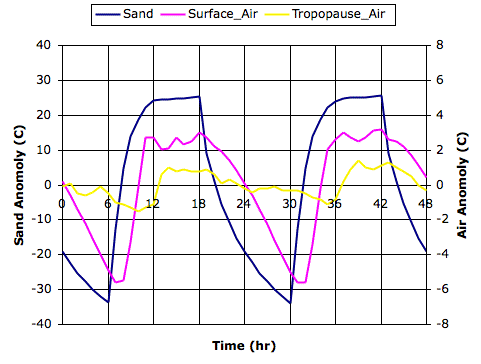
The temperature of the surface sand varies by almost 50°C, dropping as low as −12°C just before dawn. The temperature of the air a hundred meters above the sand, at the center of the bottom row of gas cells, varies by 9°C, dropping as low as 23°C. The tropopause responds far less to the day-nigh cycle, with a variation of only 2°C. These results are consistent with our observations of the desert, which we discussed at length in our Surface Cooling posts.
When we turn off the back-radiation in our simulation, the temperature of the surface sand drops by another 50°C at night, in a manner reminiscent of the Moon. And so we conclude that our atmosphere, by means of back-radiation, keeps us warm at night.
During the day, most of the heat passing into the surface gas cells does so by convection. But convection occurs only when the gas above is cooler than the surface below. In our discussion of atmospheric inversion we saw how the ground can be colder than the surface air at night, which can lead to a pocket of cold air sitting near the ground, with warmer air up above. In our simulation, we set the convection transfer to zero when the surface is colder than the surface gas.
At night, therefore, a sandy surface will radiate its heat into space, and receive no warmth from the sun. But it will receive warmth from the atmosphere, in the form of the back-radiation we described in our previous post. During the day, we found that our surface gas was radiating 226 W/m2 down to the surface. This radiation will slow the cooling of the surface at night.
We ran our simulation with Cycle heating on a sandy planet surface, 700 W/m2 solar heat during the day, daylight fraction set to 0.50, convection coefficient 20 W/m2, and transparency fraction 0.50. You will find the equilibrium state of the cell array at midnight stored in a text file here. The following graph shows the average temperature of the sand blocks, the surface gas cells, and the tropopause gas cells during two complete day-night cycles. We plot the deviation of each temperature from its average value during the cycles, which is why we call the plots "anomalies".

The temperature of the surface sand varies by almost 50°C, dropping as low as −12°C just before dawn. The temperature of the air a hundred meters above the sand, at the center of the bottom row of gas cells, varies by 9°C, dropping as low as 23°C. The tropopause responds far less to the day-nigh cycle, with a variation of only 2°C. These results are consistent with our observations of the desert, which we discussed at length in our Surface Cooling posts.
When we turn off the back-radiation in our simulation, the temperature of the surface sand drops by another 50°C at night, in a manner reminiscent of the Moon. And so we conclude that our atmosphere, by means of back-radiation, keeps us warm at night.
Thursday, July 28, 2011
Simulated Planet Surface
Our Circulating Cells Version 8.3 simulates the planet surface with a row of blocks. You can download the latest code here. These blocks can be sand or water. If they are sand, they have low heat capacity and warm up quickly in the Sun's light. If they are water, they have high heat capacity and warm up slowly. By clicking upon one of the blocks, we change it from one type to the other.
In the long run, we will implement evaporation from the water blocks, but for now we concentrate upon the heat exchange between the surface and the atmosphere. In previous versions of our program, we allowed sunlight to pass directly into the surface gas cells. Now we allow sunlight to pass all the way through the entire atmosphere to be absorbed at a solid or liquid surface. Real water reflects 4% of short-wave radiation, and sand reflects something like 10%, but our simulated water and sand absorbs all short-wave radiation.
Both sand and water are near-perfect emitters of long-wave radiation, so we allow our surface blocks to radiate heat according to Stefan's Law. We also allow heat to leave the surface blocks by convection. We determine the convection heat loss by multiplying the temperature difference between the surface and the gas by a convection coefficient. This heat leaves the surface block and enters the gas cell above.
As we described in our previous post, our simulated atmosphere is partially-transparent, to an extent specified by the transparency fraction, τ. The gas above a surface block absorbs a fraction 1−τ of the block's radiation, and the remainder passes out into space. We add the absorbed heat to the gas cell above the surface block.
Now we come to a component in the heat exchange between the atmosphere and the surface that we have not simulated or calculated before. The atmosphere itself will radiate heat downwards towards the surface, and the surface, being a perfect absorber of such radiation, will absorb all of it. The heat radiated downward by the atmosphere is often called back-radiation or downward long-wave radiation. To calculate the back-radiation, we use the same equation we applied to the tropopause in our previous post. When the transparency fraction is 0.5, the gas radiates half as much heat as a black body at the same temperature.
We ran our simulation with Day heating of 350 W/m2, all surface blocks made of sand, convection coefficient 20 W/m2K, and transparency fraction 0.50. The following figure shows the equilibrium state of the array. The surface blocks are color-coded for temperature in the same way as the gas cells, but they have an orange border to mark them as sand. A water cell has a blue border. You will find the equilibrium state of the array saved as a text file here.
As in our previous simulations, the temperature drop from the surface gas cells to the tropopause gas cells is very close to 50 K. The sandy surface settles to 303.0 K, at which temperature it radiates 480 W/m2. Of this, 240 W/m2 passes directly into space and 240 W/m2 is absorbed by the gas above. The tropopause settles to 249.0 K and radiates 110 W/m2 into space. The total radiation into space is 350 W/m2, which is the amount that is arriving from the sun, so our planet is in thermal equilibrium.
The 110 W/m2 radiated by the tropopause must pass up through the atmosphere by convection. The surface gas is at an average temperature of 298.5 K, radiating 226 W/m2 back to the surface. Thus a net 14 W/m2 passes from the surface to the atmosphere by radiation. The remaining 96 W/m2 passes into the atmosphere by convection at the surface. The surface is on average 4.5 K warmer than the gas, for which we expect only 90 W/m2 to flow by convection. But the gas cells warm by roughly 8 K while they sit upon the sand. When they first arrive from above, they are almost 10 K cooler, and 200 W/m2 flows into them by convection. Just before they rise up, they are only 2 K cooler, at which point only 40 W/m2 flows into them. When we get the program to print out the convection rate, we obtain an average of 96 W/m2, so all the heat from the Sun is accounted for.
We note that almost all the heat flowing from the surface to the atmosphere is carried by convection. The heat radiated by the surface and absorbed by the atmosphere is nearly balanced by the radiation returned by this same atmosphere. The difference is only 14 W/m2, compared to 96 W/m2 passing into the gas by convection. In our next post, we will simulate night and day over a sandy desert and see if we come up with reasonable variations in temperature at the sandy surface, the air above the surface, and the air high up in the tropopause.
In the long run, we will implement evaporation from the water blocks, but for now we concentrate upon the heat exchange between the surface and the atmosphere. In previous versions of our program, we allowed sunlight to pass directly into the surface gas cells. Now we allow sunlight to pass all the way through the entire atmosphere to be absorbed at a solid or liquid surface. Real water reflects 4% of short-wave radiation, and sand reflects something like 10%, but our simulated water and sand absorbs all short-wave radiation.
Both sand and water are near-perfect emitters of long-wave radiation, so we allow our surface blocks to radiate heat according to Stefan's Law. We also allow heat to leave the surface blocks by convection. We determine the convection heat loss by multiplying the temperature difference between the surface and the gas by a convection coefficient. This heat leaves the surface block and enters the gas cell above.
As we described in our previous post, our simulated atmosphere is partially-transparent, to an extent specified by the transparency fraction, τ. The gas above a surface block absorbs a fraction 1−τ of the block's radiation, and the remainder passes out into space. We add the absorbed heat to the gas cell above the surface block.
Now we come to a component in the heat exchange between the atmosphere and the surface that we have not simulated or calculated before. The atmosphere itself will radiate heat downwards towards the surface, and the surface, being a perfect absorber of such radiation, will absorb all of it. The heat radiated downward by the atmosphere is often called back-radiation or downward long-wave radiation. To calculate the back-radiation, we use the same equation we applied to the tropopause in our previous post. When the transparency fraction is 0.5, the gas radiates half as much heat as a black body at the same temperature.
We ran our simulation with Day heating of 350 W/m2, all surface blocks made of sand, convection coefficient 20 W/m2K, and transparency fraction 0.50. The following figure shows the equilibrium state of the array. The surface blocks are color-coded for temperature in the same way as the gas cells, but they have an orange border to mark them as sand. A water cell has a blue border. You will find the equilibrium state of the array saved as a text file here.

As in our previous simulations, the temperature drop from the surface gas cells to the tropopause gas cells is very close to 50 K. The sandy surface settles to 303.0 K, at which temperature it radiates 480 W/m2. Of this, 240 W/m2 passes directly into space and 240 W/m2 is absorbed by the gas above. The tropopause settles to 249.0 K and radiates 110 W/m2 into space. The total radiation into space is 350 W/m2, which is the amount that is arriving from the sun, so our planet is in thermal equilibrium.
The 110 W/m2 radiated by the tropopause must pass up through the atmosphere by convection. The surface gas is at an average temperature of 298.5 K, radiating 226 W/m2 back to the surface. Thus a net 14 W/m2 passes from the surface to the atmosphere by radiation. The remaining 96 W/m2 passes into the atmosphere by convection at the surface. The surface is on average 4.5 K warmer than the gas, for which we expect only 90 W/m2 to flow by convection. But the gas cells warm by roughly 8 K while they sit upon the sand. When they first arrive from above, they are almost 10 K cooler, and 200 W/m2 flows into them by convection. Just before they rise up, they are only 2 K cooler, at which point only 40 W/m2 flows into them. When we get the program to print out the convection rate, we obtain an average of 96 W/m2, so all the heat from the Sun is accounted for.
We note that almost all the heat flowing from the surface to the atmosphere is carried by convection. The heat radiated by the surface and absorbed by the atmosphere is nearly balanced by the radiation returned by this same atmosphere. The difference is only 14 W/m2, compared to 96 W/m2 passing into the gas by convection. In our next post, we will simulate night and day over a sandy desert and see if we come up with reasonable variations in temperature at the sandy surface, the air above the surface, and the air high up in the tropopause.
Wednesday, July 20, 2011
Simulated CO2 Doubling
In Planetary Greenhouse we considered an atmosphere transparent to some long-wave radiation and opaque to others. In Circulating Cells Version 8.2, which you can download here, we simulate such an atmosphere with the new transparency fraction parameter.
For the moment, we ignore the temperature difference that must exist between the planet surface and the lower atmosphere (see Surface Cooling, Part VI). We assume that the solid or liquid surface beneath one of the bottom gas cells will be at the same temperature as the gas itself. Ever since our Earth Radiator post, we have assumed that the surface of a planet is a black body radiator. If a bottom cell is at 300 K, the surface below will be at 300 K also, and by Stefan's Law it will radiate 460 W/m2. At 303 K, the radiated power increases to 480 W/m2. Black-body radiation increases as the fourth power of the temperature, so a 1% increase in temperature causes a 4% increase in radiated power.
The absorption spectrum of the Earth's atmospheric layers varies in a complex and dramatic way with wavelength, as you can see here. What made our Total Escaping Power calculation so complicated was that we had to deal with the partial absorption of some wavelengths by each atmospheric layer, and therefore the partial emission of these same wavelengths by the same atmospheric layers. We want to avoid the complexity of partial absorption at a given wavelength, so we assume that the gas in our CC8 simulation has a spectrum that vacillates between perfect transparency to perfect opacity, and does so every fraction of a micron on the wavelength scale. As a result of this vacillation, a gas cell will absorb none of the radiation at a particular wavelength, or all of it. When we look at the fraction of black-body radiation that passes through a cell, this fraction is a constant property of the cell, regardless of its temperature or pressure. We call it the transparency fraction in the CC8 program, and here we will call it τ.
By the principle of radiative symmetry, each gas cells will radiate heat at the same wavelengths it absorbs. But because all the cells around it have the same absorption spectrum, none of the heat radiated by a cell will escape into space, except for the heat radiated by the cells in the top row, which we call our tropopause. These cells radiate directly into space. The power they radiate is the power that a black body would radiate, multiplied by 1−τ.
The incoming heat from the Sun, meanwhile, passes straight through our atmosphere, because we assume that the gas is perfectly transparent to short-wave radiation. The Sun's heat warms the planet surface, which for the moment we assume is something like sand. The sand heats up rapidly until it is losing heat by radiation and convection at the same rate it is gaining heat by absorption of the Sun's light. Some heat it radiates directly into space. The rest passes into the lower atmosphere and must be transported up to the tropopause by convection, where it is radiated into space.
Bottom Gas Cell Temperature = TB
Planet Surface Temperature = TS = TB
Stefan's Constant = σ = 5.7 × 10−8 W/m2/K4
Emitted Surface Radiation = σ(TS)4
Escaping Surface Radiation = τσ(TS)4
Top Gas Cell Temperature = TT
Emitted Tropopause Radiation = σ(1-τ)(TT)4
We set τ=0.50 and ran CC8 with Day heating and the Sun's power set to 350 W/m2. After ten million iterations we are confident that we have reached equilibrium, and we obtain this array. The average temperature of the bottom cells is 301.1 K (28°C) and of the top cells is 252.1 K (−21°C). The heat radiated by the surface is 468.5 W/m2, of which 234.2 W/m2 escapes directly into space. That leaves 115.7 W/m2 of the Sun's heat to be transported up through the atmosphere. The heat radiated by the tropopause is 115.1 W/m2, leaving 0.7 W/m2 unaccounted for, which is well within the range of our rounding errors and the random fluctuations in our cell temperatures.
In our previous post, we ran our simulation for an opaque atmosphere, which corresponds to τ=0.00, and the surface temperature rose to 355 K (59°C). We see that τ=0.50 allows the surface to cool by 31°C to 28°C. In With 660 ppm CO2, we showed that doubling the CO2 concentration in the Earth's atmosphere will cause a 2% drop in the total escaping power. So now we set τ=0.49, so as to cause a 2% drop in the power escaping directly from our simulated planet surface into space. We arrive at a this array, in which the surface has warmed by 0.9°C to 302.0 K and the tropopause has warmed by 0.4°C to 252.5 K.
As a check, we run with τ=1.00, in which case the atmosphere is perfectly transparent and the surface radiates all its heat directly into space. The surface cools to 280 K (7°C). The atmosphere assumes the dry adiabatic lapse profile. But now we turn on the cell mixing by setting the mixing fraction to 0.2, and after a few hundred thousand iterations we see the entire atmosphere warm up to 280 K. This is the warm atmosphere we described in our original Greenhouse Effect post and again in Adiabatic Magic. When the atmosphere is not transporting heat to the tropopause, there is no greenhouse effect, and the atmosphere mixes until it arrives at a uniform temperature equal to the surface temperature.
We see that CC8 can simulate the effect of changing the concentration of a greenhouse gas like CO2, simply by making small changes to its transparency fraction. Once we have included evaporation, clouds, and rain into our simulation, we will be able to estimate the effect of changes in CO2 upon the average temperature of our planet surface.
For the moment, we ignore the temperature difference that must exist between the planet surface and the lower atmosphere (see Surface Cooling, Part VI). We assume that the solid or liquid surface beneath one of the bottom gas cells will be at the same temperature as the gas itself. Ever since our Earth Radiator post, we have assumed that the surface of a planet is a black body radiator. If a bottom cell is at 300 K, the surface below will be at 300 K also, and by Stefan's Law it will radiate 460 W/m2. At 303 K, the radiated power increases to 480 W/m2. Black-body radiation increases as the fourth power of the temperature, so a 1% increase in temperature causes a 4% increase in radiated power.
The absorption spectrum of the Earth's atmospheric layers varies in a complex and dramatic way with wavelength, as you can see here. What made our Total Escaping Power calculation so complicated was that we had to deal with the partial absorption of some wavelengths by each atmospheric layer, and therefore the partial emission of these same wavelengths by the same atmospheric layers. We want to avoid the complexity of partial absorption at a given wavelength, so we assume that the gas in our CC8 simulation has a spectrum that vacillates between perfect transparency to perfect opacity, and does so every fraction of a micron on the wavelength scale. As a result of this vacillation, a gas cell will absorb none of the radiation at a particular wavelength, or all of it. When we look at the fraction of black-body radiation that passes through a cell, this fraction is a constant property of the cell, regardless of its temperature or pressure. We call it the transparency fraction in the CC8 program, and here we will call it τ.
By the principle of radiative symmetry, each gas cells will radiate heat at the same wavelengths it absorbs. But because all the cells around it have the same absorption spectrum, none of the heat radiated by a cell will escape into space, except for the heat radiated by the cells in the top row, which we call our tropopause. These cells radiate directly into space. The power they radiate is the power that a black body would radiate, multiplied by 1−τ.
The incoming heat from the Sun, meanwhile, passes straight through our atmosphere, because we assume that the gas is perfectly transparent to short-wave radiation. The Sun's heat warms the planet surface, which for the moment we assume is something like sand. The sand heats up rapidly until it is losing heat by radiation and convection at the same rate it is gaining heat by absorption of the Sun's light. Some heat it radiates directly into space. The rest passes into the lower atmosphere and must be transported up to the tropopause by convection, where it is radiated into space.
Bottom Gas Cell Temperature = TB
Planet Surface Temperature = TS = TB
Stefan's Constant = σ = 5.7 × 10−8 W/m2/K4
Emitted Surface Radiation = σ(TS)4
Escaping Surface Radiation = τσ(TS)4
Top Gas Cell Temperature = TT
Emitted Tropopause Radiation = σ(1-τ)(TT)4
We set τ=0.50 and ran CC8 with Day heating and the Sun's power set to 350 W/m2. After ten million iterations we are confident that we have reached equilibrium, and we obtain this array. The average temperature of the bottom cells is 301.1 K (28°C) and of the top cells is 252.1 K (−21°C). The heat radiated by the surface is 468.5 W/m2, of which 234.2 W/m2 escapes directly into space. That leaves 115.7 W/m2 of the Sun's heat to be transported up through the atmosphere. The heat radiated by the tropopause is 115.1 W/m2, leaving 0.7 W/m2 unaccounted for, which is well within the range of our rounding errors and the random fluctuations in our cell temperatures.
In our previous post, we ran our simulation for an opaque atmosphere, which corresponds to τ=0.00, and the surface temperature rose to 355 K (59°C). We see that τ=0.50 allows the surface to cool by 31°C to 28°C. In With 660 ppm CO2, we showed that doubling the CO2 concentration in the Earth's atmosphere will cause a 2% drop in the total escaping power. So now we set τ=0.49, so as to cause a 2% drop in the power escaping directly from our simulated planet surface into space. We arrive at a this array, in which the surface has warmed by 0.9°C to 302.0 K and the tropopause has warmed by 0.4°C to 252.5 K.
As a check, we run with τ=1.00, in which case the atmosphere is perfectly transparent and the surface radiates all its heat directly into space. The surface cools to 280 K (7°C). The atmosphere assumes the dry adiabatic lapse profile. But now we turn on the cell mixing by setting the mixing fraction to 0.2, and after a few hundred thousand iterations we see the entire atmosphere warm up to 280 K. This is the warm atmosphere we described in our original Greenhouse Effect post and again in Adiabatic Magic. When the atmosphere is not transporting heat to the tropopause, there is no greenhouse effect, and the atmosphere mixes until it arrives at a uniform temperature equal to the surface temperature.
We see that CC8 can simulate the effect of changing the concentration of a greenhouse gas like CO2, simply by making small changes to its transparency fraction. Once we have included evaporation, clouds, and rain into our simulation, we will be able to estimate the effect of changes in CO2 upon the average temperature of our planet surface.
Thursday, March 24, 2011
Rotating Greenhouse
The Rotating Greenhouse configuration of Circulating Cells, Version 5 simulates the cycle of day and night that results from the rotation of a Planetary Greenhouse with respect to its sun. The day_length_hr parameter gives the length of the day-night cycle in hours. In Simulation Time, we concluded that one iteration of our simulation corresponded to one second of planetary time, so one hour is 3600 iterations and one twenty-four hour day is 86400 iterations. The sun will shine upon our Rotating Greenhouse for a fraction of the day given by day_fraction. You will find both parameters in CC5's configuration array.
As in the Planetary Greenhouse, the surface is something like sand. It heats up quickly and warms the bottom cells of the atmosphere. Now that the planet is rotating, the heat from the sun increases from zero at dawn to a maximum at mid-day, and decreases to zero again at sunset. We use a sinusoidal profile for the rise in solar heating during the day. For the entire night, no heat arrives at all. In the middle of the day, heat arrives from the sun at a rate π × Q_heating. When day_fraction is one half (the days and nights are of equal length), the average rate at which the bottom cells are warmed by the sun is Q_heating. But when the day is shorter, the average warming is less than Q_heating, and when the day is longer, the average warming is greater. For now, we leave the fraction at one half.
The top cells, meanwhile, radiates heat into space just as they did for the Planetary Greenhouse. Top cells cool by Q_heating per second when they are at temperature T_balance. Their cooling rate increases as the fourth power of their temperature.
We allow the Rotating Greenhouse simulation to run. The report line gives us the time in hours instead of the iteration counter. In the top-left of the report window we see the current rate of solar heating, in units of K/hr. After many days, the cycle of day and night reaches equilibrium, and we see the following each day.
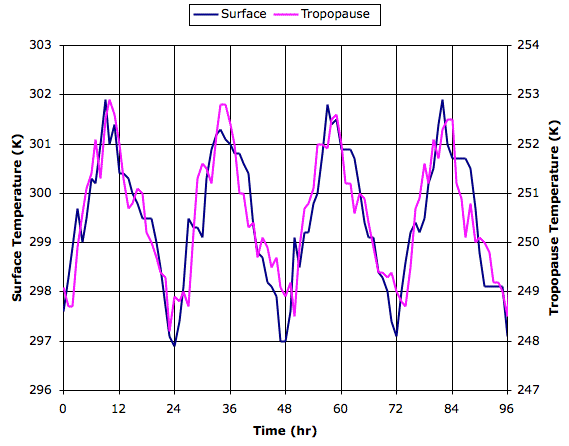
During the day, the surface cells warm as they absorb the Sun's heat. Once they warm by a few degrees, they rise, as we describe in Impetus for Circulation. The average temperature of the surface rows rises by a few degrees during the day, but not more. Our simulation does not allow surface cells to cool. And yet the average temperature of the surface row drops by a few degrees at night. When we mark a few cells near the surface, and watch them move after night falls, we see the surface cells being replaced by cooler cells from above. It is air descending from above that cools the surface row. And so our simulation confirms the mechanism for surface cooling we described in Surface Cooling, Part III.
Our simulation does not, however, show the sudden ten-degree cooling we observe standing in the desert just after the sun sets. Understanding this sudden drop was the motivation behind our Surface Cooling series of posts. Next time, we will use CC5 in its Surface Heating configuration to simulate the transport of heat within one or our large surface cells, and so obtain an estimate of how much the first twenty meters of the atmosphere will warm up during the day.
PS. You will find the array data corresponding to the start of the day with Q = 0.001 K and no cell mixing in RGH_Q001
As in the Planetary Greenhouse, the surface is something like sand. It heats up quickly and warms the bottom cells of the atmosphere. Now that the planet is rotating, the heat from the sun increases from zero at dawn to a maximum at mid-day, and decreases to zero again at sunset. We use a sinusoidal profile for the rise in solar heating during the day. For the entire night, no heat arrives at all. In the middle of the day, heat arrives from the sun at a rate π × Q_heating. When day_fraction is one half (the days and nights are of equal length), the average rate at which the bottom cells are warmed by the sun is Q_heating. But when the day is shorter, the average warming is less than Q_heating, and when the day is longer, the average warming is greater. For now, we leave the fraction at one half.
The top cells, meanwhile, radiates heat into space just as they did for the Planetary Greenhouse. Top cells cool by Q_heating per second when they are at temperature T_balance. Their cooling rate increases as the fourth power of their temperature.
We allow the Rotating Greenhouse simulation to run. The report line gives us the time in hours instead of the iteration counter. In the top-left of the report window we see the current rate of solar heating, in units of K/hr. After many days, the cycle of day and night reaches equilibrium, and we see the following each day.

During the day, the surface cells warm as they absorb the Sun's heat. Once they warm by a few degrees, they rise, as we describe in Impetus for Circulation. The average temperature of the surface rows rises by a few degrees during the day, but not more. Our simulation does not allow surface cells to cool. And yet the average temperature of the surface row drops by a few degrees at night. When we mark a few cells near the surface, and watch them move after night falls, we see the surface cells being replaced by cooler cells from above. It is air descending from above that cools the surface row. And so our simulation confirms the mechanism for surface cooling we described in Surface Cooling, Part III.
Our simulation does not, however, show the sudden ten-degree cooling we observe standing in the desert just after the sun sets. Understanding this sudden drop was the motivation behind our Surface Cooling series of posts. Next time, we will use CC5 in its Surface Heating configuration to simulate the transport of heat within one or our large surface cells, and so obtain an estimate of how much the first twenty meters of the atmosphere will warm up during the day.
PS. You will find the array data corresponding to the start of the day with Q = 0.001 K and no cell mixing in RGH_Q001
Sunday, March 13, 2011
Summary to Date
On the side-bar, you will see a new link, Summary to Date. The new page gives a history of the posts on this site. It attempts to explain what we have achieved so far, what we are working on now, and what we hope to achieve in the future. I'll maintain the page, adding paragraphs every few months. With any luck, the page will allow newcomers to make sense of what's going on.
Thursday, February 10, 2011
Planetary Greenhouse Simulation
Suppose we have a planet whose atmosphere is perfectly transparent to short-wave radiation from the Sun. The surface of the planet is made of sand, which warms up quickly. It warms until it loses heat by radiation and convection as fast as it receives heat from the Sun. Meanwhile, we suppose that the lower atmosphere is opaque to long-wave radiation so all the heat from the Sun ends up in the lower atmosphere. The lower atmosphere gets hot. It rises. Like all atmospheres, this one gets thinner as we ascend. Eventually it is so thin that it becomes transparent to long-wave radiation. This layer of the atmosphere is the tropopause. The tropopause radiates heat into space according to Stefan's Law. Air in the tropopause cools until it becomes so dense that it sinks towards the surface. And so atmospheric convection begins. The heat arriving from the Sun passes into the lower atmosphere, is carried up to the tropopause by convection, and is radiated into space.
We have described such a planet and atmosphere before, in Planetary Greenhouse. Today's CC3 program simulates the transportation of heat through such an atmosphere. You can download the program here. Running in its Planetary Greenhouse configuration, CC3 supplies heat to cells in the bottom row at a constant rate. Each bottom cell heats up by Q_heating K per iteration, regardless of its temperature. Meanwhile, a cell in the top row at temperature T_balance cools by Q_heating K per iteration. At other temperatures, the cooling rate of the upper cells increases as the fourth power of the cell temperature.

In the simulation, Q_heating represents the heat arriving from the Sun. The top cells represent the tropopause, and the heat they lose is the heat radiated by the tropopause into space. The heat radiated into space by the top cells will be equal to the heat arriving from the Sun when the temperature of all the top cells is T_balance. We start the simulation with all cells at 250 K, Q_heating = 0.01 K, and T_balance = 250 K. The top cells start to cool immediately. The lower cells start to heat up.
In CC3, we corrected our implementation of heat mixing in accordance with the suggestions made by Michele. You will find our new implementation in the heat routine. We added a reporting text window, as you can see in the screen shot above. You open the window with the Reporting button. The program calls report every reporting_interval iterations, or whenever you press the Report button. We instructed report to print the average temperature of the cell rows. We added a check procedure that checks for convergence of the simulation, as requested by Peter. In the comments below I'll provide some convergence code that you can paste into the check routine. In order to accommodate the planetary greenhouse temperature variations, we doubled the breadth of the color-coded temperature range. Red is now 320 K and blue is 220 K. The Configure button allows you to change the simulation parameters while the simulation is running. Be careful not to enter invalid numbers or else the program will abort with an error.
We're running the program now. It takes several hundred thousand iterations to reach equilibrium. In our next post, will present the vertical temperature profile under various conditions. We are curious to see how the temperature of the surface is affected by the amount of heat the atmosphere must transport, and by the amount of mixing we allow between cells.
We have described such a planet and atmosphere before, in Planetary Greenhouse. Today's CC3 program simulates the transportation of heat through such an atmosphere. You can download the program here. Running in its Planetary Greenhouse configuration, CC3 supplies heat to cells in the bottom row at a constant rate. Each bottom cell heats up by Q_heating K per iteration, regardless of its temperature. Meanwhile, a cell in the top row at temperature T_balance cools by Q_heating K per iteration. At other temperatures, the cooling rate of the upper cells increases as the fourth power of the cell temperature.

In the simulation, Q_heating represents the heat arriving from the Sun. The top cells represent the tropopause, and the heat they lose is the heat radiated by the tropopause into space. The heat radiated into space by the top cells will be equal to the heat arriving from the Sun when the temperature of all the top cells is T_balance. We start the simulation with all cells at 250 K, Q_heating = 0.01 K, and T_balance = 250 K. The top cells start to cool immediately. The lower cells start to heat up.
In CC3, we corrected our implementation of heat mixing in accordance with the suggestions made by Michele. You will find our new implementation in the heat routine. We added a reporting text window, as you can see in the screen shot above. You open the window with the Reporting button. The program calls report every reporting_interval iterations, or whenever you press the Report button. We instructed report to print the average temperature of the cell rows. We added a check procedure that checks for convergence of the simulation, as requested by Peter. In the comments below I'll provide some convergence code that you can paste into the check routine. In order to accommodate the planetary greenhouse temperature variations, we doubled the breadth of the color-coded temperature range. Red is now 320 K and blue is 220 K. The Configure button allows you to change the simulation parameters while the simulation is running. Be careful not to enter invalid numbers or else the program will abort with an error.
We're running the program now. It takes several hundred thousand iterations to reach equilibrium. In our next post, will present the vertical temperature profile under various conditions. We are curious to see how the temperature of the surface is affected by the amount of heat the atmosphere must transport, and by the amount of mixing we allow between cells.
Monday, November 8, 2010
Venus
UPDATE: Initial version of this post used diatomic gas equation for the compression of CO2. Have now corrected this oversight, and find that our estimate of Venus's surface temperature is much improved.
One of our readers suggested we consider the atmosphere of Venus. What a good idea. Let's see how well we can estimate Venus's surface temperature using our understanding of atmospheric convection and the greenhouse effect.
According to Wikipedia, Venus's tropopause is at an altitude of 65 km with temperature 243 K (−30°C) and pressure 10 kPa. The surface pressure, meanwhile, is almost a thousand times greater: 9,300 kPa, which is ninety-three times the surface pressure on Earth. Venus's atmosphere is made up almost entirely of CO2, but also contains 150 ppm of SO2 (sulfur dioxide), and this SO2 condenses into liquid droplets so that the atmosphere below the tropopause is filled with pale yellow clouds.
Venus reflects 90% of incident sunlight (it's Bond Albedo is 0.90). The remaining 10% is absorbed. We're not sure what fraction of the Sun's light reaches the surface of Venus, but our guess is 1%. The SO2 clouds refract and reflect light like our own clouds, but SO2 is a pale yellow liquid, not a clear liquid, and will absorb sunlight eventually.
According to Schriver et al., even a 0.5-μm film of SO2 ice will absorb over 20% of long-wave radiation, so a 10-μm droplet of SO2 liquid will absorb it all. The clouds of Venus are near-perfect absorbers of long-wave radiation, and near-perfect radiators too, just like our own water clouds. Unlike the Earth, however, Venus is always entirely covered with clouds. Neither the planet surface nor the lower atmosphere has any opportunity to radiate heat directly into space. Venus radiates heat directly into space only from its cloud-tops and upper atmosphere.
Although most of the sun's heat is reflected back into space, 10% is absorbed, and we estimate that around 1% reaches the surface of the planet. This heat will raise the temperature of the surface until it forces convection. Here is the convection diagram from our Atmospheric Convection post.
Gas warms at the surface. It rises, expands, radiates heat into space, falls, contracts, and warms again. As the gas expands, it gets cooler. As it contracts, it gets warmer. In our simplistic analysis, we assumed that the expansion and contraction were adiabatic, meaning they took place without any heat entering or leaving the gas. In reality, the gas radiates heat to nearby gas, absorbs heat from nearby gas, mixes with nearby gas, and generates heat through viscous friction. But our adiabatic assumption allowed us to estimate the temperature changes using the equation of adiabatic expansion and contraction. For an ideal diatomic gas, such as N2 or O2, p−0.4T1.4 remains constant during adiabatic changes, where p is pressure and T is temperature. For CO2, however, the equation is p−0.3T1.3 at 300 K and p−0.2T1.2 at 1300 K (see here for thermodynamic properties of CO2).
The Earth's tropopause is at altitude 15 km. According to our typical conditions, the tropopause is at 220 K with pressure 22 kPa, while the surface pressure is 100 kPa. Adiabatic compression implies that air descending from the tropopause to the surface will heat up to 340 K. But the Earth's surface is at only 280 K. Air descending 10 km from the tropopause to the surface appears to lose 20% of its heat while contracting to a third of its original volume.
The atmosphere of Venus will heat up as it descends from the tropopause to the surface, and it will be hottest if it does not lose any heat on the way down. The pressure rises from 10 kPa in the tropopause to 9,300 kPa at the surface. The temperature starts at 240 K. Adiabatic compression of an ideal diatomic gas would heat the gas to 1700 K. If we use p−0.25T1.25 as an approximation for CO2, we estimate a final temperature of 941 K.
According to Wikipedia, the surface temperature of Venus is actually 740 K. Carbon dioxide falling 65 km from the tropopause to the surface appears to lose 20% of its heat while being compressed into less than a hundredth of its original volume.
Despite the 20% difference between our calculations and our observations, we see that atmospheric convection makes the surface of Venus incredibly hot while leaving the surface of the Earth delightfully temperate.
If we recall our post on work by convection and another on atmospheric dissipation, we see that a strong convection cycle causes powerful weather. Venus's convection cycle produces an order of magnitude more contraction and expansion than the Earth's. We expect Venus's weather to be an order of magnitude more extreme. And indeed it is.
One of our readers suggested we consider the atmosphere of Venus. What a good idea. Let's see how well we can estimate Venus's surface temperature using our understanding of atmospheric convection and the greenhouse effect.
According to Wikipedia, Venus's tropopause is at an altitude of 65 km with temperature 243 K (−30°C) and pressure 10 kPa. The surface pressure, meanwhile, is almost a thousand times greater: 9,300 kPa, which is ninety-three times the surface pressure on Earth. Venus's atmosphere is made up almost entirely of CO2, but also contains 150 ppm of SO2 (sulfur dioxide), and this SO2 condenses into liquid droplets so that the atmosphere below the tropopause is filled with pale yellow clouds.
Venus reflects 90% of incident sunlight (it's Bond Albedo is 0.90). The remaining 10% is absorbed. We're not sure what fraction of the Sun's light reaches the surface of Venus, but our guess is 1%. The SO2 clouds refract and reflect light like our own clouds, but SO2 is a pale yellow liquid, not a clear liquid, and will absorb sunlight eventually.
According to Schriver et al., even a 0.5-μm film of SO2 ice will absorb over 20% of long-wave radiation, so a 10-μm droplet of SO2 liquid will absorb it all. The clouds of Venus are near-perfect absorbers of long-wave radiation, and near-perfect radiators too, just like our own water clouds. Unlike the Earth, however, Venus is always entirely covered with clouds. Neither the planet surface nor the lower atmosphere has any opportunity to radiate heat directly into space. Venus radiates heat directly into space only from its cloud-tops and upper atmosphere.
Although most of the sun's heat is reflected back into space, 10% is absorbed, and we estimate that around 1% reaches the surface of the planet. This heat will raise the temperature of the surface until it forces convection. Here is the convection diagram from our Atmospheric Convection post.

Gas warms at the surface. It rises, expands, radiates heat into space, falls, contracts, and warms again. As the gas expands, it gets cooler. As it contracts, it gets warmer. In our simplistic analysis, we assumed that the expansion and contraction were adiabatic, meaning they took place without any heat entering or leaving the gas. In reality, the gas radiates heat to nearby gas, absorbs heat from nearby gas, mixes with nearby gas, and generates heat through viscous friction. But our adiabatic assumption allowed us to estimate the temperature changes using the equation of adiabatic expansion and contraction. For an ideal diatomic gas, such as N2 or O2, p−0.4T1.4 remains constant during adiabatic changes, where p is pressure and T is temperature. For CO2, however, the equation is p−0.3T1.3 at 300 K and p−0.2T1.2 at 1300 K (see here for thermodynamic properties of CO2).
The Earth's tropopause is at altitude 15 km. According to our typical conditions, the tropopause is at 220 K with pressure 22 kPa, while the surface pressure is 100 kPa. Adiabatic compression implies that air descending from the tropopause to the surface will heat up to 340 K. But the Earth's surface is at only 280 K. Air descending 10 km from the tropopause to the surface appears to lose 20% of its heat while contracting to a third of its original volume.
The atmosphere of Venus will heat up as it descends from the tropopause to the surface, and it will be hottest if it does not lose any heat on the way down. The pressure rises from 10 kPa in the tropopause to 9,300 kPa at the surface. The temperature starts at 240 K. Adiabatic compression of an ideal diatomic gas would heat the gas to 1700 K. If we use p−0.25T1.25 as an approximation for CO2, we estimate a final temperature of 941 K.
According to Wikipedia, the surface temperature of Venus is actually 740 K. Carbon dioxide falling 65 km from the tropopause to the surface appears to lose 20% of its heat while being compressed into less than a hundredth of its original volume.
Despite the 20% difference between our calculations and our observations, we see that atmospheric convection makes the surface of Venus incredibly hot while leaving the surface of the Earth delightfully temperate.
If we recall our post on work by convection and another on atmospheric dissipation, we see that a strong convection cycle causes powerful weather. Venus's convection cycle produces an order of magnitude more contraction and expansion than the Earth's. We expect Venus's weather to be an order of magnitude more extreme. And indeed it is.
Subscribe to:
Posts (Atom)
