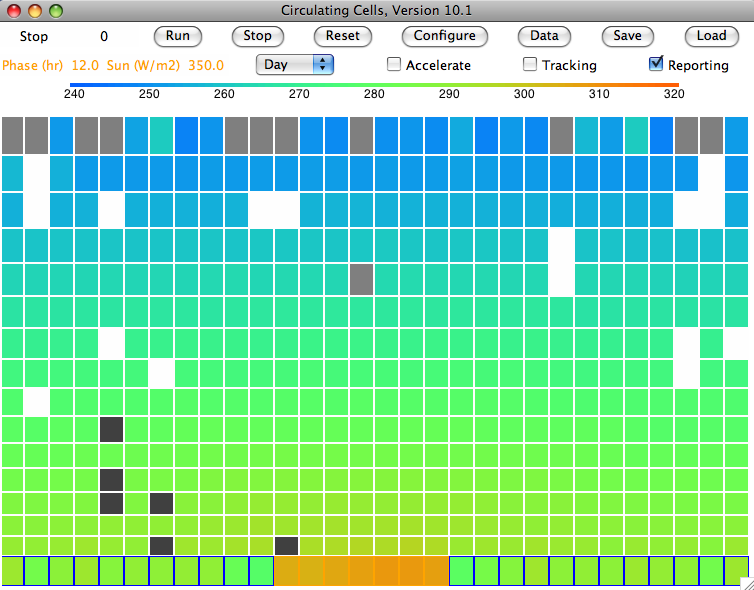
We are going to add to our
Circulating Cells simulation the absorption and emission of
long-wave radiation by clouds. As we showed
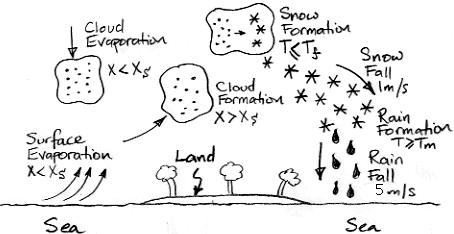
earlier, a liquid water depth of 100 μm absorbs over 99% of all long-wave radiation. Rain contains liquid water also, and ice is a good absorber of long-wave radiation too. We will add the
equivalent depth of snow, rain, and cloud droplets for each cell, and so obtain the depth of water within the cell that acts to absorb long-wave radiation.
We note that the same addition of rain, snow, and cloud droplets does not apply to the transmission of
short-wave radiation. Water is transparent to short-wave radiation, and clouds reflect it by
refracting it through millions of microscopic droplets. But rain and snow contain thousands of times fewer drops and crystals for a given depth of water, so they are thousands of times less effective at refracting sunlight.
For simplicity, we will assume the water in a cell is either transparent or opaque to long-wave radiation, but not in-between. If the combined concentration of rain, snow, and cloud droplets in a cell is greater than
wc_opaque, we will assume the entire gas cell is opaque to long-wave radiation. Otherwise, the cell will absorb long-wave radiation as if it were dry, as determined by our
transparency_fraction. With our 300-kg cells, a concentration of 0.33 g/kg corresponds to 100 μm of water.
Now we are faced with the possibility of multiple layers of cloud, snow, and rain, all absorbing and emitting long-wave radiation in all directions. The first simplification we make is to assume each gas cell radiates only vertically upwards and downwards. Because our columns of cells are much the same as one another on average, the net effect of this simplification will be small. Even with this simplification, we see that a cloud can absorb radiation from a cloud below, and emit radiation back to that same cloud below, and upwards to a third cloud.
We will calculate the effect of long-wave radiation in the following way. We start at the surface and allow it to radiate as a
black body. We allow this upward radiation to enter the first gas cell. We calculate how much is absorbed by the cell and how much keeps going. We calculate how much power the gas cell itself radiates upwards. We add this to the existing upward radiation. We move on to the cell above, and so on, until we get to the tropopause. At the tropopause, we assume the atmosphere above is transparent to long-wave radiation, so all upward-going radiation passes out into space.
We repeat the same process, going down. We start with the tropopause gas cell in each column and move down cell by cell until we arrive at the bottom, at which point all the downward-going radiation is absorbed by the surface. We first considered this kind of downward-going long-wave radiation in our
Back Radiation post. It is distinct from the solar radiation that penetrates the atmospheric clouds because it is radiation emitted by the clouds, rain, snow, and atmospheric gas themselves.
In any cell, the long-wave radiation going up is the
upwelling radiation and the long-wave radiation going down is the
downwelling radiation. At the tropopause, the upwelling radiation is the heat leaving our planetary system. It is our
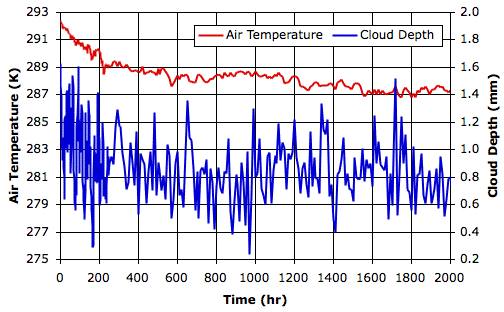
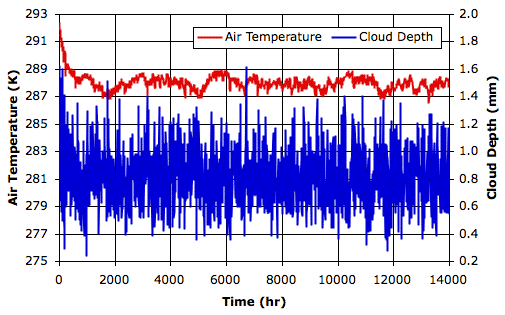
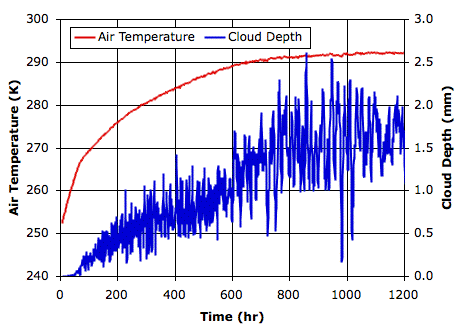
total escaping power. When our simulation converges to equilibrium, we should find that the average solar power penetrating to the surface is equal to the average total escaping power.