When a block of four cells in our
atmospheric simulation rotates by convection, the center of mass of the block drops. In
Impetus for Convection, we called the work done by the descending block
buoyancy work. When we calculated buoyancy work with our
CC5 program, we considered only the movements of the four cells within the rotating block. Today we present a calculation of buoyancy work that takes account of the work required to move the cells above the block, and we find that our original calculation over-estimated buoyancy work by more than a factor of three.
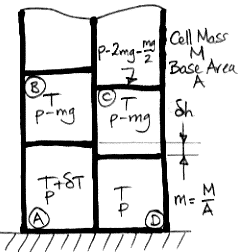
We start with a block of four cells that has no tendency to circulate, warm up the lower-left cell, and consider the buoyancy work this single warm cell can generate for us. Here is the diagram we presented in our
Buoyancy post. The cell on the lower left is warmer by δ
T than the adiabatic profile we derived
earlier. We use R for the specific gas constant, Cv for the specific heat capacity at constant volume, and Cp for the specific heat capacity at constant pressure. As always, we have Cp=R+Cv for an ideal gas.
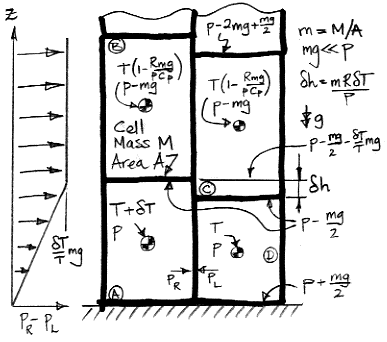
When a cell expands adiabatically from
p to
p−
mg, its temperature drops from
T to
T(1−
mg/
p)
R/Cp. When
mg≪
p, this expression simplifies to
T(1−R
mg/
pCp), which is why the upper cells in our diagram have temperature
T(1−R
mg/
pCp).
As we heat cell
A by δ
T, we raise its center of mass by δ
h/2 and the center of mass of cell
B by δ
h. We also raise the gas above
B by δ
h. Cells
C and
D do not move, nor does the gas above them. The weight of each cell is
m per unit base area, and the weight of the gas above cell
B is the pressure in cell
B, which is
p−
mg. As we supply heat to
A to warm it up, the work done by the expanding cell against gravity is W1.
W1 = mgδ
h + mgδ
h/2 + (
p−
mg)δ
h =
mRδ
T(1 +
mg/2
p)
We now allow the four cells to rotate one step clockwise. Cells
C and
D move to the bottom. They are at temperature
T. Cell
B will be on the upper-right, with its temperature unchanged at
T(1−R
mg/
pCp). Cell
A, having expanded adiabatically from pressure
p to
p−
mg, will cool down, but will still be warmer than cell
B by δ
T ' = δ
T(1−R
mg/
pCp).
Suppose we remove heat from cell
A until it is the same temperature as
B. Once it has cooled by δ
T ', the block of four cells will be identical to the one we started with before we heated
A by δ
T. As
A cools by δ
T ', its center of mass drops by δ
h'.
δ
h' =
mRδ
T '/(
p−
mg) =
mRδ
T(1−R
mg/
pCp)/(
p−
mg)
We note that
mg≪
p and this allows us to simplify the above expression.
δ
h' ≈
mRδ
T(1 +
mgCv/
pCp)/
p
As cell
A shrinks, its own center of mass drops by δ
h'/2 and the gas above drops by δ
h'. Now we can calculate the work done by gravity,
W2, as the cell cools.
W2 =
mgδ
h'/2 + (
p−
mg)δ
h' = δ
h'(
p−
mg/2) =
mRδ
T(1 +
mgCv/
pCp)(1−
mg/2
p)
Once again we note that
mg≪
p, which allows us to simplify our expression.
W2 ≈
mRδ
T(1 +
mgCv/
pCp −
mg/2
p)
When we heated cell
A by δ
T, we increased the gravitational potential energy of the cells and upper gas by and amount W1. When we allowed the cells to rotate, they produced buoyancy work, the size of which we would like to determine. When we allowed cell
A to cool, we decreased the gravitational potential energy of the cells and upper gas by an amount W2, and in doing so we returned these gases to their original state. By conservation of energy, the difference between W1 and W2 must be equal to the buoyancy work.
W1−W2 =
mRδ
T(R
mg/
pCp)
The buoyancy work per kilogram of gas in the four cells,
Wb, is therefore:
Wb =
mgR
2δ
T/4
pCp
We interpret our expression for
Wb as follows. When the cells rotate, cell
B drops by δ
h, which does work
mgδ
h =
mgRδ
T/
p. But at the same time, all the cells above
A rise by δ
h'−δ
h>0, and this requires work
mgRCvδ
T/
pCp. The buoyancy work is the work done by
B as it drops, minus the work required to raise the upper gas, divided by the mass of the four cells combined, which gives us the expression for Wb above. In our
CC5 calculation, we ignored the work required to raise the upper gas, and so over-estimated the buoyancy work by a factor of Cp/R, which is 3.5 for air.
If cell
B contains 300 kg/m
2 of air at 100 kPa and 300 K, and δ
T for cell
A is 10°C, the buoyancy work is 6.2 J/kg, which is sufficient to accelerate the cells to 3.5 m/s. These cells would be 250 m high, so at 3.5 m/s they would circulate in one minute. Using our old calculation of buoyancy work, the circulation would take almost twice as long.