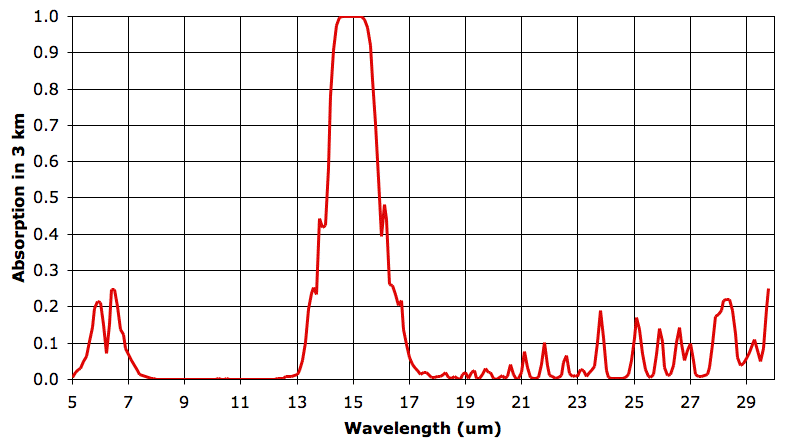
As before, we say our 3-km layer is transparent at a particular wavelength when it absorbs less than 63% of that wavelength, and opaque otherwise. The fifth 3-km layer is transparent from 5.0 to 14.2 μm and from 15.8 to 30 μm.
Between the fourth and fifth layer of the Earth's atmosphere, water vapor concentration drops from 100 ppm to 10 ppm. Absorption by water vapor almost disappears. So far as our transparent-opaque simplification is concerned, the absorption by water vapor does indeed disappear. The atmosphere becomes transparent to all long-wave radiation except for 14.2 to 15.8 μm. These wavelengths are absorbed by CO2. Unlike water vapor concentration, CO2 concentration remains constant as we ascend from 0 km to 12 km.
The concentration of C02 may be constant, but the amount of CO2 in each cubic meter of air is still decreasing. The density of the air decreases as we ascend. Thus the absorption by CO2 in the fifth layer is less intense than in the fourth layer. The CO2 in the fourth 3-km layer absorbs radiation from 14.1 to 15.9 μm. By radiative symmetry, these are the same wavelengths that the fourth layer itself radiates up to the fifth 3-km layer. Of this radiation, only the narrow ranges 14.1 to 14.2 μm and 15.8 to 15.9 μm will pass through the fifth layer and out into space. The remaining 90% will be absorbed by CO2 in the fifth layer.

No comments:
Post a Comment